BioCardia (NSDQ:BCDA) said today it filed for a second FDA investigational device exemption to launch a trial of its CardiAMP cell therapy for treating chronic myocardial ischemia and refractory angina as it seeks a second indication. The San Carlos, Calif.-based company’s CardiAMP therapy uses autologous cells to treat chronic myocardial ischemia, and is designed to stimulate […]
Regenerative Medicine
These lab-grown blood vessel replacements could benefit dialysis patients
University of Minnesota researchers have developed a blood vessel replacement made of biological materials in a lab. The lab-grown vessels have no living cells at implantation and could be used as a graft for kidney dialysis patients. The lab-engineered blood vessel replacement is the first nonsynthetic, decellularized graft that is repopulated with cells using the […]
Histogenics inks deal to develop Neocart for the Japanese market
Histogenics (NSDQ:HSGX) has inked a deal with Medinet to develop and commercialize its Neocart implant for the Japanese market. The Waltham, Mass.-based company’s Neocart implant is designed to repair knee cartilage damage. The product is made using a patient’s own cells, harvested from the surface of the patient’s femur. “This transformative collaboration for Histogenics is […]
Researchers reverse diabetes in mice by infusing them with blood stem cells
Researchers at Boston Children’s Hospital successfully reversed Type I diabetes in a mouse model using blood stem cells, according to a study published this week in Science Translational Medicine. The researchers used cells that were pre-treated to make more of a protein, PD-L1, which is deficient in mice and people with Type I diabetes. “There’s really […]
Orthocell wins CE Mark for CelGro device
Orthocell (ASX:OCC) said today that its CelGro collagen medical device won CE Mark approval for a range of dental bone and soft tissue regeneration procedures. The company said it’s in talks with the strategic commercial partners to distribute its product throughout Europe and other key regions. Orthocell is also reportedly holding discussions with key opinion leaders […]
Avita Medical aims for $13m to bring ReCell burn injury device to U.S.
Avita Medical (ASX:AVH) said today that it hopes to raise $13.2 million to fund its efforts to bring its ReCell autologous cell harvesting device for treatment of burn injuries to the U.S. The Australia-based company said it received commitments from investors for a $3.5 million private placement. Avita added that it plans to undertake a rights […]
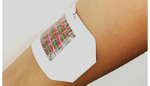
Smart bandage releases medication as needed
A team of researchers from the University of Nebraska, Harvard Medical School and MIT have created a smart bandage that can be loaded with antibiotics, painkillers or other drugs and triggered by a smartphone. The bandage, made of electrically conductive fibers individually coated in a drug-loaded hydrogel, could be used to deliver multiple drugs at […]
MiMedx closes Stability Biologics sale
MiMedx (NSDQ:MDXG) said this week that it completed the divestiture of its subsidiary, Stability Biologics, back to the former stockholders of Stability, Inc. The deal, which closed on Sept. 30, included a promissory note issued by Stability Biologics for $3.5 million in favor of MiMedx. “When we signed the definitive agreement for the divestiture, we communicated […]
FDA expands protocol for investigational ReCell burn injury device
Avita Medical (ASX:AVH) said today that the FDA approved a supplement to the company’s investigational device exemption for its ReCell autologous cell harvesting device. The approved treatment protocol for burn injuries has been simplified, according to the company, and the number of approved investigational sites has grown from eight to 15. Up until now, treatment using […]
Avita Medical submits PMA app for ReCell burn injury treatment
Avita Medical (ASX:AVH) said today that it submitted its pre-market approval application to the FDA for its ReCell autologous cell harvesting device. The company’s system is designed to reduce the amount of skin harvesting needed to treat burn injuries compared to conventional treatments. The PMA application is supported by clinical data from two trials. The […]