
Ocular Therapeutix wrapped up patient enrollment for a Phase II trial evaluating the company’s OTX-DP tear duct plug, the company announced this week.
By drugdelivery

Ocular Therapeutix wrapped up patient enrollment for a Phase II trial evaluating the company’s OTX-DP tear duct plug, the company announced this week.
By drugdelivery
By drugdelivery
By drugdelivery


By drugdelivery
By drugdelivery

Federal regulators this week published a final rule governing Current Good Manufacturing Practice requirements for products made up of a combination of devices, drugs and/or biologics, categories which are usually regulated by separate FDA divisions.
The long-awaited rule is meant to provide clarification on CGMP requirements for products that include any fusion of the medical products either as packaged together or as a "single-entity" combination.
By drugdelivery

Hampton, N.J.-based healthcare company Ikaria won FDA clearances for an upgraded software package and 3 non-invasive respiratory care devices for use with its Inomax DSIR drug delivery system.
The Inomax DS and DSIR deliver the company’s proprietary Inomax vasodilator solution, which the company calls the only FDA-approved drug for treatment of infant hypoxic respiratory failure, associated with pulmonary hypertension.
By drugdelivery

Medical device company Delcath Systems (NSDQ:DCTH) is on a roll, notching a couple new regulatory milestones this month for its organ-isolating ChemoSat chemotherapy treatment systems.
By drugdelivery

Delcath Systems (NSDQ:DCTH) won expanded CE Mark approval in the European Union for its ChemoSat organ-isolating cancer treatment system, landing indication for a new drug used to target liver tumors.
By drugdelivery
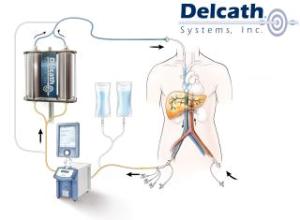
The FDA agreed to review Delcath Systems’ (NSDQ:DCTH) new drug application, bringing the device maker 1 step closer to launching its organ-isolating chemotherapy system on the U.S. market.

