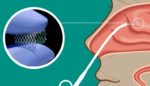
Lyra Therapeutics (NSDQ:LYRA) announced today that it initiated the Phase 3 Enlighten I trial for its LYR-210 treatment. Watertown, Massachusetts-based Lyra’s Phase 3 Enlighten I clinical trial will evaluate LYR-210, delivered through Lyra’s proprietary XTreo drug-eluting implant, in adult, surgically naïve chronic rhinosinusitis (CRS) patients. The company designed LYR-210 for delivery in a brief, non-invasive, […]
MIT team working on all-in-one diabetes management devices
MIT researchers are touting a platform aimed at helping patients maintain healthy glucose levels by streamlining the diabetes management process. The MIT researchers say it’s hard for people with diabetes to maintain a treatment regimen. Typically at every meal, they need to estimate carbohydrate content, draw blood to measure blood glucose levels, and calculate the […]
Fractyl Health touts preliminary data from diabetes reversal study
Fractyl Health this week announced encouraging interim feasibility data from its diabetes reversal technology study. Lexington, Massachusetts–based Fractyl’s Revita-T2Di long-term open-label cohort assessed eight patients followed through at least four weeks having been treated. The company’s Revita system resurfaces the upper intestine lining (duodenal mucosa) to treat metabolic disease in patients with insulin-treated type 2 […]
FDA grants IDE approval for Capillary Biomedical’s 7-day-wear infusion set
Capillary Biomedical announced today that it received FDA investigational device exemption (IDE) for its SteadiFlow infusion technology. Irvine, California-based Capillary Biomedical designed its new SteadiFlow seven-day-wear infusion set technology to significantly extend patient wear time to a week and maintain insulin stability. The SteadiSet infusion set, powered by the SteadiFlow technology platform, features an integrated […]
Ahead of G7 launch, Dexcom CEO plans for ‘more activity than we’ve ever seen’ in 2022
With a major product approval and launch in its sights, Dexcom prepares for a big year in 2022. Across 2021, Dexcom (NSDQ:DXCM) took several steps forward. The company touted its next-generation G7 continuous glucose monitor (CGM) and announced a significant regulatory nod and partnership for real-time application programming interfaces (APIs), to list a couple. As […]
FDA grants new drug approval to Glenmark’s rhinitis-treating nasal spray
Glenmark Pharmaceuticals announced today that it received FDA approval for a new drug application (NDA) for Ryaltrais. Ryaltris, a fixed-dosed, prescription drug product nasal spray treats symptoms of seasonal allergic rhinitis in adults and pediatric patients over 12 years of age in the U.S. Glenmark’s fully owned subsidiary, Glenmark Specialty S.A. (Switzerland), received the NDA […]
Fort HealthCare, Ivenix partner on infusion safety effort with smart pump
Ivenix announced today that it successfully implemented its smart infusion pump at the Fort HealthCare hospital in Wisconsin. Boston-based Ivenix designed its Ivenix Infusion System, a combination of a large-volume infusion pump and infusion management software, to offer unique capabilities within the infusion therapy market, with pumps deployed at Fort HealthCare set to connect with […]
Roche launches its connected blood glucose meter in Europe
Roche announced today that it launched its Cobas Pulse system in select countries accepting CE mark approval in Europe. Basel, Switzerland–based Roche designed its Cobas Pulse system to combine the form factor of a high-performance blood glucose meter with simple usability and expanded digital capabilities comparable to that of a smartphone. The platform joins Roche’s […]
Dali Medical Devices completes first implementation of SAN-Light passive safety needle
Dali Medical Devices announced today that it completed the first commercial implementation of the SAN-Light passive safety needle. Tel Aviv, Israel-based Dali designed the SAN-Light single-use, sterile, hypodermic safety needle for compatibility with any Luer-lock syringe for subcutaneous or intramuscular drug administration. SAN-Light makes up part of Dali’s SAN (safe auto needle) family of safety […]
Glytec partners with Roche to enhance insulin dosing decision support software
Glytec announced today that it entered into a strategic partnership with Roche Diagnostics USA for a digital health collaboration. Waltham, Massachusetts-based Glytec’s partnership combines its FDA-cleared Glucommander insulin dosing decision support software with Roche’s expertise in medical devices and IT solutions in an effort to address the challenges associated with inpatient blood sugar management at […]