Shares in Dipexium Pharmaceutical (NSDQ:DPRX) dived 84% today after it reported that its OneStep-1 and OneStep-2 Phase III clinical trials of Locilex in patients with mild infections of diabetic foot ulcers did not meet its primary or secondary clinical endpoints. Both trials failed to show any meaningful difference in wound closure rate between the Locilex arm and the […]
Clinical Trials
Imprimis touts ‘dropless therapy’ study for cataract surgery
Imprimis Pharmaceuticals (NSDQ:IMMY) touted data today from a peer-reviewed study of patients who received its “dropless therapy” following cataract surgery. The San Diego-based company uses a compounded antibiotic and steroid formulation in single, injectable doses administered by physicians following ocular surgery. The treatment is designed to eliminate the need for patient-administered eye drops, potentially eliminating patient […]
aTyr Pharma wins FDA fast track for muscular dystrophy therapy
aTyr Pharma (NSDQ:LIFE) said today that its candidate Resolaris was granted fast track designation by the FDA for the treatment of facioscapulohumeral muscular dystrophy, making it the 1st therapeutic candidate to receive the designation for the rare form of muscular dystrophy. Resolaris is derived from a naturally occurring protein released by human skeletal muscle cells. aTyr is […]
FDA delays Inovio phase III trial of DNA immunotherapy candidate
Inovio Pharmaceuticals (NSDQ:INO) said today that the FDA placed a hold on its proposed Phase III clinical program for its investigational DNA immunotherapy candidate designed to treat pre-cancers and cancers caused by human papillomavirus. The California-based company completed a randomized, double-blind Phase II trial of the candidate in July 2014, evaluating the DNA immunotherapy treatment in women […]
Mylan, Theravance touts Phase III data for COPD drug
Mylan (NSDQ:MYL) and Theravance Biopharma, Inc. (NSDQ:TBPH) said today that revefenacin, the 1st nebulized bronchodilator in development for the treatment of chronic obstructive pulmonary disease, demonstrated positive results in 2 replicate phase III efficacy studies. Data from more than 1,250 moderate to severe COPD patients showed that both studies met their efficacy and safety endpoints. Revefenacin […]
Akesys, Elixir Medical tout 1st implant for Prava bioresorbable peripheral stent
Akesys Medical and Elixir Medical Corporation said today that it implanted its 1st Prava sirolimus eluting bioresorbable peripheral scaffold for the treatment of blockages in the superficial femoral artery. The scaffold was developed to keep the large artery in the thigh open and restore blood flow to the leg and resorb into the body, making […]
FDA awards product development grants for rare diseases
The FDA said yesterday that it awarded 21 clinical trial research grants totaling more than $23 million over the next 4 years to stimulate product development for patients with rare diseases. The Orphan Products Clinical Trials Grants Program has awarded more than $370 million to fund more than 590 clinical studies since its creation in […]
Intersect ENT’s Resolve sinus implant meets endpoints in pivotal trial
Intersect ENT (NSDQ:XENT) said today that its Resolve sinus implant met both primary endpoints in the Resolve II pivotal trial. The Menlo Park, Calif.-based company’s Resolve steroid-releasing sinus implant is a minimally invasive treatment for patients with recurrent ethmoid sinus obstruction. The 300-patient Resolve II trial compared the bilateral Resolve implant with a sham procedure. The study evaluated the change […]
Novocure enrolls 1st patient in Phase III brain metastases trial
Novocure (NSDQ:NVCR) said that the 1st patient has been enrolled in a Phase III pivotal trial of its tumor-treating field technology in combination with radiosurgery to treat brain metastases from non-small-cell lung cancer. The St. Helier, N.J.-based company’s tumor treating fields (TTFields) are low-intensity, intermediate-frequency, alternating electric fields designed to disrupt cell division within cancer cells. Novocure’s commercial product, Optune, […]
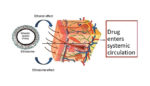
Report finds ethosomes as promising transdermal delivery mechanism
Ethosomes – nanocarriers loaded with ethanol – can penetrate the skin’s protective layers and deliver a variety of drugs, a review published in Current Clinical Pharmacology reported today. Transdermal delivery of drugs is touted by some researchers as promising, since it’s noninvasive and avoids any interaction with the gastrointestinal tract. Ethosomes are malleable and their […]