Mylan (NSDQ:MYL) defended chairman Robert Coury to Institutional Shareholder Services, an shareholder advisory group, earlier this week as the firm prepares to release a report advising shareholders how they should vote in the upcoming re-election of Coury and other directors. Major investors like the New York City and State pension funds have urged shareholders to vote against […]
Drug-Device Combinations
Clearside Biomedical highlights preliminary data from retreatment trial
Clearside Biomedical (NSDQ:CLSD) touted preliminary data from a non-interventional and retrospective trial of patients who participated in the completed Phase II trial of CLS-TA for suprachoroidal administration. Clearside’s formulation of triamcinolone acetonide, in combination with intravitreally administered Eylea, was evaluated as a treatment for macular edema associated with retinal vein occlusion. The company’s Phase II […]
Novocure launches Optune, chemotherapy combo trial for glioblastoma
Novocure (NSDQ:NVCR) said today that the 1st patient was enrolled in a Phase II pilot trial evaluating Optune with bevacizumab, a chemotherapy, for patients with refractory recurrent glioblastoma. In recurrent GBM, Optune and bevacizumab are approved as monotherapies. Novocure’s consortium study is slated to evaluate the efficacy and safety of the Optune-chemo combo in patients […]
Surefire Medical touts infusion tech in liver cancer trial
Surefire Medical touted data today from a retrospective analysis of 85 bridge-to-transplant liver cancer patients treated with transarterial chemoembolization. The data showed that therapy delivered using the Surefire infusion system yielded an 80% complete response in 1 treatment compared to 52% with a standard endhole microcatheter. The analysis also demonstrated a lower recurrence rate of […]
ivWatch raises $14.8m for infusion therapy tech
ivWatch said today that it closed a $14.8 million financing round to support the company’s direct go-to-market initiatives and strategic partners activities. Undisclosed angel investors, family funds and a major hospital system invested in the round, according to the company. “The support ivWatch has seen from our investors and partners has been incredible. They are […]
Amaranth Medical looks to recapture the potential of bioresorbable scaffolds
Dr. Antonio Colombo had finished his presentation of 9-month data for Amaranth Medical‘s Aptitude sirolimus-eluting bioresorbable scaffold when an audience member asked about adverse events. He told the EuroPCR attendee that they hadn’t seen any adverse events related to the device during the trial – and that’s unusual. Bioresorbable scaffolds, like Abbott‘s (NYSE:ABT) Absorb, have been […]
Glytec highlights hospital cost-savings for insulin therapy
At the 6th International Hospital Diabetes meeting, Glytec‘s eGlycemic Management System was featured in several studies demonstrating the products’s ability to save hospitals money. One case study presented at the meeting described a health system that adopted Glytec’s eGlycemic Management System and saw 1st-year cost savings of $7.5 million. The Kaweah Delta Medical Center attributed the […]
Biopolymer tested as long-acting glucose control therapy
Researchers from Duke University have developed a biopolymer that could provide weeks of glucose control for patients with diabetes – in a single injection. The controlled-release treatment lasted for weeks in primates, according to a study published in Nature Biomedical Engineering. The biomedical engineers suggested that this new, injectable solution could replace daily or weekly insulin shots for […]
Artificial pancreas startup lands funding support from T1D Exchange
Admetsys, a startup developing artificial pancreas tech for hospitals and surgical care, won an investment from T1D Exchange. The terms of the financing were not disclosed. A collaboration between the 2 groups is part of T1D Exchange’s initiative to support the development of automated insulin delivery technology, according to the organization. Hospitalizations can be challenging for […]
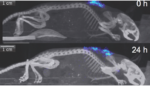
Living Cell Technologies NTCell meets primary endpoint in Phase I/IIa Parkinson’s trial
Living Cell Technologies (ASX:LCT) touted data today showing its NTCell cell therapy is safe 130 weeks after treatment in a Phase I/IIa Parkinson’s trial. The study’s primary endpoint was safety. The trial, which enrolled and treated 4 patients, involved implanting 40 NTCell capsules into 1 side of the brain. The company reported that treatment with […]