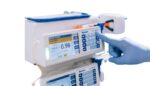
Tandem Diabetes Care (Nasdaq:TNDM) announced today that it launched Tandem Source, a diabetes management platform. The platform, for both insulin pump users and healthcare providers, brings together features of Tandem Diabetes Care’s legacy offerings. It combines t:connect, t:connect HCP and t:connect Portal with new, comprehensive data reporting in one central, scalable platform. All Tandem pump […]
Featured
Report: There’s still ‘mutual interest’ with EOFlow, Medtronic despite deal falling through
Business Korea reports that EOFlow CEO Kim Jae-jin is leaving the door open with regard to the company’s collapsed deal with Medtronic (NYSE:MDT). Medtronic announced this week that it terminated its $738 million agreement to acquire EOFlow, a maker of insulin patch pumps. The medtech giant cited “multiple breaches” of terms in the agreement as […]
Beta Bionics integrates iLet bionic pancreas with Dexcom G7
Beta Bionics announced today that it began the U.S. launch of its iLet bionic pancreas with the Dexcom G7 continuous glucose monitor (CGM). The launch marks the second G7-compatible automated insulin delivery system to hit the market in as many days. Yesterday, Tandem Diabetes Care became the first to launch an artificial pancreas using Dexcom’s […]
Medtronic nixes $738M deal for insulin patch pump maker EOFlow
Medtronic (NYSE:MDT) filed a Form 8K with the SEC today stating that it terminated its agreement to buy EOFlow. The medtech giant struck a $738 million deal to acquire the Seongnam, South Korea-based insulin patch pump maker in May. EOFlow develops the EOPatch — a tubeless, wearable and fully disposable insulin delivery device. Medtronic’s SEC filing […]
Tandem Diabetes Care launches first automated insulin pump integrated with Dexcom G7 CGM
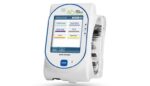
Tandem Diabetes Care (Nasdaq:TNDM) announced today that it launched its updated t:slim X2 insulin pump software with the Dexcom (Nasdaq:DXCM) G7. Integration makes Tandem the first to offer automated insulin delivery with the latest-generation Dexcom continuous glucose monitor (CGM). The FDA cleared the next-generation Dexcom G7 in December 2022. By February of this year, the […]
BD recalls more Alaris pumps due to compatibility issues with Cardinal Health syringes
The FDA determined that another recall of BD (NYSE:BDX) Alaris infusion pumps is Class I, the most serious kind. In the latest Alaris recall, the company cites compatibility issues with Cardinal Health Monoject syringes. The issue mainly relates to changes made to the products by Cardinal Health. It affects more than 1 million total devices. […]
Aptar wins FDA contract to study low global warning potential metered dose inhalers
Aptar Pharma announced today that it entered into a contract with the FDA to work on low global warming potential (low-GWP) inhalers. The FDA enlisted Aptar to study the challenges of developing low-GWP propellant metered dose inhalers (MDIs). The Crystal Lake, Illinois-based company expects its study to help define the potential target product profile of […]
Baxter issues Novum IQ infusion pump warning
Correction: the original version of this article identified this as another recall for Novum IQ. Baxter issued a notification related to a Nov. 15 recall, not a notification for a separate recall. This story has been updated. Baxter (NYSE:BAX) issued an urgent medical device correction notice regarding its Novum IQ syringe infusion pump. A few […]
Eitan Medical’s Sapphire infusion pump recall is Class I
The FDA determined that the recall of certain Eitan Medical Sapphire infusion pumps is Class I, the most serious kind. Netanya, Israel-based Eitan recalled certain Sapphire pumps running software version Rev 16.10. The recall includes the Sapphire multi-therapy pump, the Sapphire Epidural pump and the Sapphire Plus pump. Eitan initiated the recall on Sept. 11, […]
FDA warning doesn’t affect Embecta syringes, company says
Embecta (Nasdaq:EMBC) says an FDA safety communication regarding certain plastic syringes has no impact on its own products. The FDA yesterday warned of the potential for device failures with plastic syringes manufactured in China. Failures could include leaks, breakage and other problems. The agency said it received information about quality issues associated with several Chinese […]