GlaxoSmithKline (NYSE:GSK) will not likely face generic competition to its lung drug Advair in the US this year. Yesterday, the FDA denied the application for a 2nd generic competitor. Hikma Pharmaceuticals (LON:HIK) disclosed today that the FDA decided not to approve its version of the inhaled asthma treatment, due to “major” problems with the application. This comes […]
Featured
Ethicon touts new anti-infection suture guidelines
Johnson & Johnson (NYSE:JNJ) subsidiary Ethicon touted updated anti-infection suture guidelines from the Centers for Disease Control and Prevention, which recommend that healthcare professionals “consider the use of triclosan-coated sutures for the prevention of SSI [surgical site infection].” Ethicon Plus Sutures are the only globally available sutures coated with triclosan that stop bacteria commonly associated with […]
OncoSec inks deal with Merck to study drug-device therapy for metastatic melanoma
OncoSec Medical (NSDQ:ONCS) said today that it inked a clinical trial collaboration and supply agreement with Merck (NYSE:MRK) to evaluate the combination of its ImmunoPulse IL-12 electroporation device with Merck’s Keytruda therapy in a Phase II clinical trial. The trial aims to evaluate the drug-device combination in patients with metastatic melanoma – specifically patients who experienced disease […]
Halo Pharma launches development & manufacturing facility for pediatric drugs
Halo Pharma launched its Pediatic Center of Excellence today to support the development and manufacturing of drugs tailored for pediatric indications. The company said it will use the manufacturing facility to continue working with pharmaceutical companies to develop pediatric dosage forms of already-approved adult drugs. When the FDA requests that a company conduct clinical trials for […]
Nobilis, Submersible Systems to develop hand-held inhalation device for neurological drugs
Nobilis Therapeutics said today that it partnered with device manufacturer Submersible Systems to develop a portable, hand-held inhalation device that administers a xenon gas-based therapeutic, NBTX-001. The companies said they are working on customizing and testing the drug-device combination. The group plans to pursue regulatory approval with the FDA, supported by upcoming clinical trials in […]
Patients with Parkinson’s disease help shape clinical trial design
Margaret Sheehan isn’t a scientist, but she is 1 of many patient scientists joining an effort to incorporate Parkinson’s patients’ preference into the design of clinical trials for new treatments. Sheehan, a Virginia-based lawyer, has had Parkinson’s disease since 2004. She told Drug Delivery Business News that when the folks at the Medical Device Innovation Consortium presented last spring […]
Mylan beats Q1 earnings by a penny, misses on revenue
Shares in Mylan (NSDQ:MYL) rose 2% in pre-market activity today after the EpiPen maker beat earnings expectations on Wall Street with its 1st quarter results. The Canonsburg, Penn.-based company posted profits of $66.4 million, or 12¢ per share, on sales of $2.72 billion for the 3 months ended March 31, for bottom-line growth of 377.7% on sales growth […]
Cerus lands $47m for Intercept red blood cell system
Cerus (NSDQ:CERS) said today that it exercised $46.6 million in additional options from its contract with the Biomedical Advanced Research & Development Authority, a part of the U.S. Dept. of Health and Human Services’ Office of the Assistant Secretary for Preparedness and Response. Concord, Calif.-based Cerus is developing the Intercept blood system, a pathogen reduction device designed […]
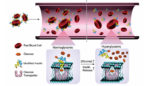
Researchers combine modified insulin and red blood cells to regulate blood sugar in diabetic mice
Researchers from North Carolina State University and the University of North Carolina at Chapel Hill have designed a glucose-responsive “smart” insulin delivery system using modified insulin and red blood cells. In an animal study of mice with Type I diabetes, the system effectively reduced blood sugar levels for 48 hours. The team’s work was published […]
OptiNose raises $37m in Series D for commercial efforts
OptiNose said today that it closed a $37 million Series D round led by Fidelity Management and Research Company. Avista Capital Partners, Entrepreneurs Fund and other current investors also contributed to the round. The Yardley, Penn.-based company said it plans to use the newly-acquired funds to support commercial development of its lead product candidate, OPN-375, […]