Migraine treatment that has been safe for adults has recently proven to be safe for use in children as well. The minimally-invasive treatment only takes a few minutes for children and teenagers to be able to feel the effects. The treatment involves a sphenopalatine ganglion (SPG) block that does not need needles. It uses a […]
Pain Management
Ocular misses estimates for Q4 earnings, but beats on rev
Shares in Ocular Therapeutix (NSDQ:OCUL) fell today after the biopharmaceutical company missed earnings expectations, but beat sales estimates on Wall Street with its 4th quarter results. The Bedford, Mass.-based company posted losses of -$-12.8 million, or -52¢ per share, on sales of $0.5 million for the 3 months ended Dec. 31, for bottom-line loss of -20.5% on […]
Pacira prices $300m senior notes offering
Pacira Pharmaceuticals (NSDQ:PCRX) said today that it priced $300 million of convertible 2.375% senior notes due 2022 in a private placement to qualified institutional buyers. The Parsippany, N.J.-based company also gave purchasers a 30-day option to buy an additional $45 million aggregate principal amount of notes. The company expects to bring in about $290 million […]
AcelRx gains on Q4 earnings
Shares in AcelRx Pharmaceuticals (NSDQ:ACRX) rose today after the pharmaceutical company met expectations on Wall Street with its 4th quarter results. The Redwood City, Calif.-based company pared its losses to -$9.7 million, or -21¢ per share, on sales of $6.4 million for the 3 months ended Dec. 31, for bottom-line growth of 8.2% on sales growth of […]
FDA accepts AcelRx’s NDA for Dsuvia pain reliever
AcelRx Pharmaceuticals (NSDQ:ACRX) said today that the FDA accepted the company’s New Drug Application for its Dsuvia pain relief therapy. The federal watchdog set a target decision date for October 12 this year. The company’s Dsuvia candidate is composed of 30 sufentanil tablets delivered sublingually using a disposable, pre-filled single-dose applicator to patients with moderate-to-severe […]
Adapt Pharma seeks regulatory nod for naloxone nasal spray in Europe
Adapt Pharma said today that it filed a European Marketing Application for its naloxone hydrochloride nasal spray for the treatment of opioid overdose. The Dublin-based company’s product is a ready-to-use, needle-free device that delivers a single dose of naloxone in an 0.1 mL nasal spray and does not require assembly prior to use, according to […]
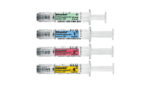
Fresenius Kabi launches Dilaudid injection in Simplist prefilled syringes
Fresenius (NYSE:FMS) business group Fresenius Kabi said today that its Dilaudid hydromorphone HCl injection is available in 4 different presentations of the Simplest prefilled syringes. The Lake Zurich-Ill. company’s Simplist syringes have a 2-year shelf life and do not require any assembly or point-of care preparation. The prefilled syringes sport different colors associated with different dose […]
AcelRx touts Dsuvia data for burn victims
AcelRx Pharmaceuticals (NSDQ:ACRX) said today that it plans to present data from a study evaluating its Dsuvia pain reliever in patients with moderate-to-severe acute pain resulting from trauma or injury. The company’s Dsuvia candidate is composed of 30 sufentanil tablets delivered sublingually using a disposable, pre-filled single-dose applicator. Dsuvia is investigational and not approved in […]
FDA accepts Flexion’s NDA for osteoarthritis steroid injection
Flexion Therapeutics (NSDQ:FLXN) said today that the FDA accepted its New Drug Application for Zilretta – its extended-release steroid injection. The Burlington, Mass.-based company won Fast Track status from the watchdog for its osteoarthritis injection in 2015. Flexion touts Zilretta as the 1st intra-articular, extended-release therapy for patients with knee osteoarthritis pain. The injection uses microsophere technology […]
West Pharmaceutical Services introduces new drug delivery safety products
West Pharmaceutical Services, an injectable drug administration technology provider, has introduced two new products meant to improve drug delivery safety. The LyoSeal cap is all-plastic and provides instant sealing for lyophilized drug products. West designed the LyoSeal to minimize risks involved with established lyophilization processes while maintaining the necessary sealing requirements. The NovaGuard SA Pro […]