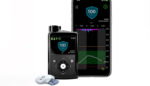
Abbott (NYSE:ABT) announced today that the FDA cleared its FreeStyle Libre 2 and FreeStyle Libre 3 for automated insulin delivery (AID) integration. AID systems help people manage daily diabetes care by automatically adjusting and administering the insulin delivered by an insulin pump based on real-time glucose data from their FreeStyle Libre 2 or FreeStyle Libre […]
Technology
Expanded Medicare coverage for CGM to go into effect next month
The Centers for Medicare & Medicaid Services today published a final local coverage determination (LCD) expanding coverage for CGMs. New, expanded coverage of continuous glucose monitors (CGMs) under Medicare go into effect in April. CMS first published the LCD modifying coverage criteria for CGMs in October. The modification includes people with diabetes who receive insulin […]
Bigfoot Biomedical wins FDA clearance for Bigfoot Unity Android app
Bigfoot Biomedical announced today that it received FDA 510(k) clearance for the Android version of its Bigfoot Unity mobile app. Milpitas, California-based Bigfoot received FDA clearance for its diabetes management platform in May 2021. Its initial rollout featured compatibility with iOS devices. The first-of-its-kind system includes connected smart pen caps designed to recommend insulin doses […]
Vivani Medical wants to bring a subdermal drug delivery implant to the diabetes market
Where some have tried and failed, Vivani Medical believes its drug-delivering implants can go the distance. When Second Sight Medical and Nano Precision Medical merged last year, the rebranded company — Vivani Medical (Nasdaq:VANI)— had a goal: bring a six-month, subdermal drug delivery implant that treats conditions including type 2 diabetes to market. CEO Dr. […]
Tandem Diabetes Care appoints new board chair
Tandem Diabetes Care (Nasdaq:TNDM) recently announced the appointment of Rebecca Robertson as the chair of its board of directors. Robertson initially joined Tandem’s board as an independent director in January 2019. She succeeds Kim Blickenstaff as chair. Blickenstaff intends to continue as a member of the board. “We’re deeply grateful to Kim for his instrumental […]
Glaukos submits FDA New Drug Application for iDose TR drug-eluting implant
Glaukos (NYSE:GKOS) announced today that it submitted a New Drug Application (NDA) to the FDA for its iDose TR implant. Aliso Viejo, California-based Glaukos designed iDose TR as a micro-invasive intraocular implant. It continuously delivers therapeutic levels of a proprietary formulation of travoprost from within the eye over extended periods of time. Once all travoprost […]
iRadimed issues urgent field safety notice for some infusion pump syringe adapters
iRadimed (Nasadq:IRMD) issued an urgent medical device correction letter to warn of issues with some infusion pump syringe adapter sets. The letter notifies customers of a potential issue with some 1057 Syringe Adapter Sets. The issue relates to the syringe venting function during use. This can lead to reduced flow with the MRidium infusion pump, […]
Study backs Medtronic next-gen MiniMed 780G insulin pump for type 1 diabetes
Medtronic (NYSE:MDT) announced today that one-year data backs the use of its next-generation MiniMed 780G automated insulin pump. The ADAPT study compared MiniMed 780 G against multiple daily injections of insulin with an intermittently scanned CGM. ADAPT, which included 82 subjects, marks the first multi-national, randomized controlled study comparing these methods. The study evaluated adults […]
Report: Apple moves forward with glucose monitoring tech
Apple reportedly believes it could shake up the CGM market with non-invasive glucose monitoring through the Apple Watch. Blooomberg reported that the company has a “moonshot-style” project in the works, according to “sources familiar with the matter.” The report said the tech giant still has years ahead in this development of the technology, which wouldn’t […]
Glooko, Sanofi partner on insulin pen connectivity
Glooko announced today that it entered into a partnership with Sanofi to integrate a connected device for insulin pens into its digital platform. SoloSmart, a single-piece, add-on connected device works with Sanofi’s SoloStar and DoubleStar insulin injection pens. It records the insulin dose, date and time of injection. Through this partnership, the company’s can offer […]