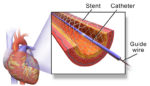
Medtronic (NYSE:MDT) said last week that its clinical study comparing the In.Pact AV drug-coated balloon to percutaneous transluminal angioplasty (PTA) met primary safety and effectiveness endpoints. The study pitted the In.Pact AV DCB against PTA in patients with de novo or non-stented restenotic arteriovenous fistulae lesions. Results were presented at the Cardiovascular and Interventional Radiology Society […]
Medtronic
Surmodics reaps $10m from Abbott on SurVeil balloon milestone
Surmodics (NSDQ:SRDX) said today that it’s due for a $10 million milestone payment from Abbott (NYSE:ABT) after closing enrollment for the Transcend pivotal trial of its SurVeil drug-coated balloon for treating peripheral artery disease. Abbott in February 2018 paid $25 million up front for the global commercialization rights to the SurVeil device, which uses a proprietary […]
Study: No difference between Biotronik, Medtronic or Boston Scientific stents
A study comparing drug-eluting stents made by Biotronik, Medtronic (NYSE:MDT) and Boston Scientific (NYSE:BSX) found no statistically significant difference on target vessel failure or stent thrombosis after three years. The 3,514-patient Bio-Resort trial compared Biotronik’s Orsiro biodegradable sirolimus-eluting stent, Medtronic’s thin-strut zotarolimus-eluting Resolute Integrity stent and the Synergy biodegradable everolimus-eluting stent. Three-year results for some 3,393 […]
Medtronic wins FDA approval for new extended-wear infusion set
Medtronic (NYSE:MDT) said it earned investigational approval from the FDA to proceed with a trial for a new extended-wear infusion set. The goal of the study is to collect clinical data that supports the use of the device for up to seven days, more than twice the amount of time any infusion set can currently […]
Medtronic bids for expanded FDA indication for Guardian Sensor 3
Medtronic (NYSE:MDT) said it asked the FDA for an expanded indication for its Guardian Sensor 3 device that would open the glucose sensor up for Medicare reimbursement. The Fridley, Minn.-based medtech titan said it filed for pre-market approval of non-adjunctive labeling for Guardian Sensor 3 for use with its MiniMed 670G insulin management system. If […]
Medtronic recalls MiniMed pump models on cybersecurity risk
Medtronic (NYSE:MDT) today recalled two models of its MiniMed insulin pumps after researchers discovered cybersecurity risks that could allow a hacker to take control of the devices. The recall affects 11 models in Fridley, Minn.-based Medtronic’s line of MiniMed 508, MiniMed Paradigm and MiniMed Paradigm Veo pumps, according to the FDA. Medtronic said the pumps […]
FDA panel wants more, better data on paclitaxel-eluting devices
An FDA advisory panel on paclitaxel-eluting devices for treating peripheral artery disease yesterday found that the mortality signal indicated by a meta-analysis last year exists but that further study is needed to determine whether it’s a class effect and to pin down the cause of death. A meta-analysis published in the Journal of the American Heart Assn. last December […]
FDA panel finds ‘smoking gun, but no bullet or dead bodies’ in paclitaxel devices
Members of an FDA advisory panel yesterday cited conflicting evidence from studies of paclitaxel-eluting devices in treating peripheral artery disease, with the panel’s chairman calling the data “a smoking gun, but no bullet or dead bodies at this particular point.” A meta-analysis published in the Journal of the American Heart Assn. last December suggested that PAD patients treated with paclitaxel-coated […]
FDA: More data needed on paclitaxel devices
More data is needed on devices that elute the drug paclitaxel to determine if they pose a higher mortality risk than non-eluting devices in treating peripheral artery disease, the FDA said ahead of an advisory panel meeting today. A meta-analysis published in the Journal of the American Heart Assn. last December suggested that PAD patients treated with paclitaxel-coated balloons […]
ADA 2019: Hybrid closed-loop devices take center stage
The American Diabetes Association’s 79th Scientific Sessions is a five-day conference where researchers and healthcare professionals share the latest in diabetes research. More than 180 sessions and over 2,000 research presentations took place during the meeting on June 7-11 in San Francisco. During the meeting, diabetes experts shared data and research from clinical trials. Here […]