 Frost & Sullivan announced today that it recognizes Becton Dickinson (NYSE:BDX) with a 2020 Global Technology Innovation Award.
Frost & Sullivan announced today that it recognizes Becton Dickinson (NYSE:BDX) with a 2020 Global Technology Innovation Award.
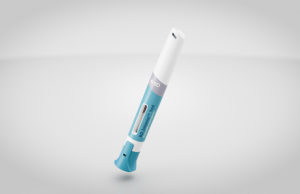
BD received the award for its Intevia autoinjector, which, as part of the company’s differentiated self-injection systems portfolio, facilitates high-viscosity biologics delivery for treating chronic diseases, according to a news release.
Intevia, a two-step, push-on-skin, handheld autoinjector delivers biologic medicines of differing viscosities and can be adapted to deliver a range of drug volumes without any need to customize the system and subsystem components.
Becton Dickinson launched the BD Intevia 1mL platform in 2019 and is currently developing a 2.25mL version as a patient-centric device to be integrated with the BD Neopak XtraFlow prefillable syringes.
The award from Frost & Sullivan for the Intevia technology recognizes a product with innovative features and functionality that is gaining rapid acceptance in the market, according to the consulting firm.
“BD Intevia’s 1-mL and 2.25-mL dose configurations meet the demand for a delivery mechanism for viscous biologics. The drug-device combination improves the self-injection experience in home settings, which, in turn, improves disease management, patient adherence, and quality of life,” Frost & Sullivan industry principal Unmesh Lal said in the release. “BD Intevia’s 1 mL design configuration integrates the control point feature that allows the autoinjector to probe the target site without getting activated, allowing patients to test the target injection site and choose the location.”
“A partner in the customer assembly and filling process, BD assists pharma customers in installing facilities for filing and assembly equipment at their production site or contract manufacturing organization. Maintaining a global footprint with manufacturing, commercial, and technical organizations across multiple geographies, BD’s breadth of capabilities cements its position as a global leader in self-injection systems.”

