CGM’s beginnings
1999: FDA approves first professional CGM
Known as a “Continuous Glucose Monitor System” from Medtronic MiniMed (Medtronic Diabetes, Northridge, Calif.), the device provided three days of glucose data meant initially for a physician rather than a patient to review. The device was intended to help physicians establish patients’ glucose profiles and monitor insulin therapy. It took patient readings every 10 seconds while providing 5-minute averages. The sensor had a maximum life of 72 hours. Like many others that would follow, the device would require fingerstick-based blood glucose calibration.
2001: FDA approves first real-time CGM
The GlucoWatch Biographer from Cygnus Inc. won marketing approval for adults with diabetes. It was the only system to provide a stream of glucose readings to patients when introduced. While the armband-based device was noninvasive, it was not widely successful because it tended to cause site irritation. Animas, which eventually acquired the rights to the GlucoWatch and its successor, the GlucoWatch G2 Biographer, stopped selling the technology in 2007. Johnson & Johnson acquired Animas in 2006.
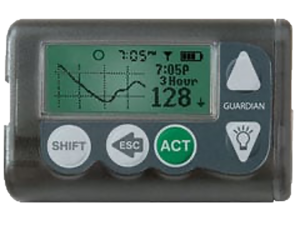
2004: Medtronic wins FDA approval for its Guardian CGM System
The Guardian CGM from the medical device giant could warn users when their blood sugar hits dangerous levels. The device provided glucose values every five minutes.
2004: Dexcom introduces its first real-time CGM
Known as the Dexcom STS (short for short-term sensor), the device shared glucose every five minutes. Like Medtronic’s Guardian device, the STS could also provide alerts when glucose levels dip or rise above predefined thresholds. The device was intended to be used for up to 72 hours.

