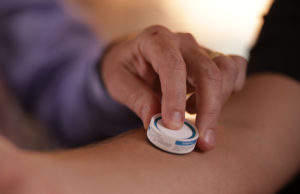
The company develops a vaccine patch based on its proprietary HD-MAP delivery technology. Vaxxas designed its HD-MAP technology to use an ultra-high-density array of micro projections, invisible to the human eye. Applied to the skin as a patch, the system rapidly delivers vaccines to immune cells below the skin surface.
HD-MAP offers improved thermostability, potentially reducing the cost and complexity of cold-chain distribution and storage. It also potentially requires less training to administer and could even lead to self-administration.
Global charitable foundation, Wellcome, made the award to conduct investigational new drug-enabling and first-in-human studies. The company expects to complete the project within two years.
It aims to accelerate the delivery of a potentially second-generation typhoid vaccine, based on SKYTyphoid, an approved typhoid conjugate. SK Bioscience, Vaxxas’ collaborator in the project, and the International Vaccine Institute developed the formulation. The developers formulated the vaccine candidate for more stability at higher temperatures than required for needle and syringe vaccination.
“We are excited to be initiating this important work with Wellcome and SK bioscience to leverage our HD-MAP vaccine platform to potentially enhance typhoid vaccination,” said David L. Hoey, President and CEO of Vaxxas. “We believe our HD-MAP can play a critical role in extending the global reach of typhoid conjugate vaccines, and make a significant difference to the lives of many.”
Vaxxas also has programs underway for its technology to deliver COVID-19 and influenza vaccines. The company raised $23 million at the end of last year as well to support its platform.

