Clearance covers people with diabetes ages six and up, expanding the Tandem portfolio of products.
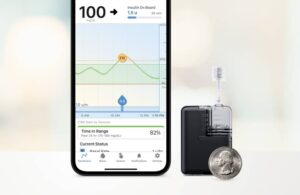
San Diego-based Tandem says Mobi, which is fully controllable from a mobile app, is the world’s smallest durable AID system.
Mobi features a 200-unit insulin cartridge and an on-pump button to provide an alternative to phone control for insulin boluses. It comes in at less than half the size of the existing Tandem pump system, the t:slim X2 pump.
According to the company, it can fit in a coin pocket, clip to clothing or go on the body with an adhesive sleeve.
“Testing the limits of pump miniaturization, Tandem Mobi joins the t:slim X2 pump in our family of insulin delivery solutions bringing new options in wearability, the flexibility to disconnect, and full phone control,” said John Sheridan, Tandem president and CEO. “Through this expansion, we are delivering on our commitment to bring greater choice, along with the proven benefits of Tandem’s technology, to more people living with diabetes.”
More features of the Tandem Mobi insulin pump tech
Tandem Mobi also comes with detachable infusion sets compatible with all existing Tandem-branded infusion sets manufactured by Convatec. This also includes a new five-inch tubing option made just for the Mobi system. The infusion sets allow users to temporarily disconnect from their pump for convenience. According to the company, they offer the flexibility of more than 30 mix-and-match infusion site and tubing length combinations.
Mobi also features Tandem’s Control-IQ technology for automated insulin delivery. Control-IQ, a hybrid closed-loop algorithm, uses compatible continuous glucose monitor (CGM) sensor values to predict glucose levels 30 minutes ahead. Every five minutes, the algorithm adjusts insulin delivery to prevent highs and lows. It still enables the user to manually bolus for meals while delivering automatic correction boluses to prevent hyperglycemia.
The mobile app control enables full iOS command through a compatible iPhone. Modern features include water-resistance, inductive charging and wireless remote software updates, too.
Tandem plans to conduct a limited release of Mobi in late 2023. The company intends to make it fully available in early 2024.
More competition added to the automated insulin delivery space
The FDA clearance of the Tandem Mobi adds one more innovative offering to people with diabetes. Over the past year or two, we’ve seen a bit of an insulin pump boom, with the t:slim X2 playing its own part.
In addition to Tandem’s offerings, both Insulet and Medtronic are making waves in automated insulin delivery.
The Insulet Omnipod 5 became the first available tubeless, wearable, automated insulin delivery system after the FDA cleared it in January 2022. In April, the company garnered FDA clearance for its Omnipod GO long-acting insulin delivery device. The new patch pump covers the basal-only insulin population. The target population typically takes daily injections of long-acting insulin.
Meanwhile, Medtronic made huge strides with the FDA approval of its MiniMed 780G automated insulin delivery system in April. Not long after, the company escalated its activity in automated insulin delivery with the planned $738 million acquisition of EOFlow.
Korea-based EOFlow develops the EOPatch, a tubless, wearable and fully disposable insulin delivery device. While EOPatch holds authorization in a number of geographies, it remains unavailable commercially in the U.S. The company submitted the insulin delivery device for U.S. FDA clearance in January.
When Medtronic announced the planned acquisition, investors reacted negatively to the likes of Insulet and Tandem, suggesting a power shift in automated insulin delivery. However, analysts quelled those reservations at the time, calling it “an overreaction.”
Another recent addition to the competitive space is the iLet ACE automated insulin pump from Beta Bionics, which received FDA clearance in May.
More remains to be seen with the latest innovation from Tandem and what the rest of the space is doing, but it’ll be an interesting area to watch.

