 This year’s flu season has been exceptionally bad – it’s already claimed the lives of dozens of kids and it’s more widespread than any other year on record, according to the Centers for Disease Control and Prevention.
This year’s flu season has been exceptionally bad – it’s already claimed the lives of dozens of kids and it’s more widespread than any other year on record, according to the Centers for Disease Control and Prevention.
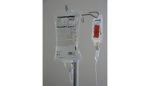
It’s also making a shortage of intravenous fluids and IV bags worse than it already is.
IV saline bags have long been in short supply, but the issue was exacerbated by Hurricane Maria’s impact on Puerto Rico and the medical device manufacturers that have facilities on the island. FDA commissioner Dr. Scott Gottlieb has been working with suppliers to get their manufacturing plants back on Puerto Rico’s electrical grid, but the devastation left behind by the hurricane has made it challenging for manufacturers to ramp up to full capacity.
Last month, the FDA chief reported that the agency’s efforts, combined with those of local lawmakers and companies like Baxter (NYSE:BAX), are proving useful. The shortage should improve the coming weeks and months, he said.
But the ill-timed flu season isn’t helping the situation.
“Although the saline shortage is improving, this year’s worse-than-normal flu season and workarounds deployed by healthcare providers in the wake of this shortage have increased demand for saline and other products,” Gottlieb noted today in prepared remarks.
Large volume saline bags, which are usually used to hydrate patients, and small volume IV bags, which are used to deliver medicine, are in short supply. Now that healthcare workers are filling empty IV containers to compound their own IV saline solutions, Gottlieb is worried that the empty containers could be next on the shortage list.
Some providers have told the FDA that there are spot shortages of certain antivirals used to combat the flu, as well as flu tests. There isn’t a nationwide shortage, Gottlieb said, but the FDA is reportedly keeping a close eye on the situation.
“Along with antivirals and device products we are also monitoring influenza vaccine supplies. While there have been some reported spot shortages, flu vaccines are still available,” he added. “I strongly encourage anyone who hasn’t had a flu shot to get one and anyone experiencing flu-like symptoms to promptly consult with their health care provider about appropriate treatment options.”
In an attempt to mitigate the IV fluid bag shortage, the FDA has extended the expiration dates of certain large volume saline bags. The agency reviewed safety data submitted by the company and determined that the product could be safely used for longer than originally determined.
Gottlieb also said the FDA is working with Baxter and B. Braun to bring products from foreign facilities into the U.S.
The flu season is expected to reach its peak soon, Gottlieb said, and the FDA chief is looking at this experience as a lesson learned.
“The hurricanes and flu season have underscored the importance of preparedness and connectivity across the agency, the industries we regulate, as well as distributors and health care providers to best serve patients that need access to these critical products,” he said.


Very helpful information shared. Keep it up.