From Bioness’s new distribution deal to InBrace’s launch of new braces technology, here are 7 medtech stories we missed this week but thought were still worth mentioning. 1. Obalon receives approval to commercialize in Middle East Obalon Therapeutics announced in a May 11 press release that it has received regulatory approvals in select Middle East […]
Eurasian governments progress on unified device regulatory system
By Stewart Eisenhart, Emergo Group Regulators in five Eurasian countries report progress in efforts to establish a harmonized medical device market that would incorporate Russia, Armenia, Belarus, Kazakhstan and Kyrgyzstan. Get the full story here at the Emergo Group’s blog. The opinions expressed in this blog post are the author’s only and do not necessarily […]
European medical device and IVD regulations officially published
By Stewart Eisenhart, Emergo Group The European Commission has officially published the Medical Device Regulations (MDR) 2017/745 and In Vitro Diagnostic Regulations (IVDR) 2017/746 on May 5, 2017 in the Official Journal of the European Union (OJEU). The MDR will replace the Active Implantable Medical Devices Directive (90/385/EEC — AIMD) and the Medical Devices Directive […]
Common audit problems: Publishing audit reports
By Stewart Eisenhart, Emergo Group In the final installment of Emergo’s blog series on medical device quality management system audit problems, we examine the audit report publication process and related challenges. Get the full story here at the Emergo Group’s blog. The opinions expressed in this blog post are the author’s only and do not […]
FDA approves first ALS treatment in over 2 decades
The FDA recently announced that it has approved the drug Radicava (edaravone) to help treat patients who have amyotrophic lateral sclerosis (ALS), also known as Lou Gehrig’s disease. ALS is a progressive neurodegenerative disease that affects the nerve cells in the brain and spinal cord, according to the ALS Association. Motor neurons in the brain […]
China FDA clarifies device recall requirements
By Stewart Eisenhart, Emergo Group Chinese medical device market regulators have made reporting forms necessary for recall management available online. Get the full story here at the Emergo Group’s blog. The opinions expressed in this blog post are the author’s only and do not necessarily reflect those of DrugDeliveryBusiness.com or its employees.
Medtech stories we missed this week: May 5, 2017
From Biomerics’s $38.5 million expansion in Salt Lake City to new study results and partnerships, here are 7 medtech stories we missed this week but thought were still worth mentioning. 1. Biomerics opening new corporate HQ Biomerics announced in a May 1 press release that it is moving its corporate headquarters in Salt Lake City. The […]
FDA and NIH release final template for clinical trial protocols
By: Peter Marks, M.D., Ph.D. A little more than a year ago, FDA and NIH announced the availability of a draft template for investigators to use when organizing clinical trial protocols, which are essentially roadmaps for conducting a clinical trial. Today, we are excited to announce that the NIH-FDA Joint Leadership Council has issued a final […]
10 new crowdfunded health devices that will intrigue you
With crowdfunding websites like Kickstarter and Indiegogo, companies can obtain the funding for ideas they want to come to life (even potato salad parties), with the promise of something in return to the donors. And while crowdfunding presents challenges for regulated medical devices, it’s proven to be a boon for startups touting potentially innovative health and […]
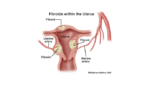
New medicines could eliminate uterine fibroid surgeries
Decisions Resources Group, a healthcare research and data company, suggests that the launch of new medicines could be a long-term management alternative to uterine fibroids surgeries over the next 10 years. Novel selective progesterone receptor modulators (SPRMs) like Allergan/Gedeon Richter’s ulipristal, Bayer’s vilaprisan and Repros’s Proellex – and gonadotropin-releasing hormone (Gn-RH) antagonists like AbbVie’s elagolix, […]