Insulet (Nasdaq:PODD) announced today that Wayde McMillan decided to leave his role as EVP, CFO and treasurer. McMillan’s departure goes into effect on Oct. 20, 2023. He intends to join 3M as CFO of its Health Care business, which the company expects to spin off late this year or early next year. President and CEO […]
Phillips-Medisize, GlucoModicum partner on needle-free CGM
Molex company Phillips-Medisize today announced a partnership with GlucoModicum to design a new continuous glucose monitor (CGM). The companies teamed up to design and eventually commercialize a proprietary, non-invasive, wearable CGM device. They aim to remove the technology and patient-care roadblocks that exist within continuous glucose monitoring. Phillips-Medisize develops drug delivery, in vitro diagnostic and […]
Abbott study says FreeStyle Libre users on insulin show lower HbA1c levels
Abbott (NYSE:ABT) today announced real-world data associating its FreeStyle Libre CGM with significant HbA1c reductions. The continuous glucose monitor (CGM) played a part in significant reductions in HbA1c, the average glucose levels over a three-month period. It was also associated with a lower rate of hospitalization for people with type 2 diabetes on both multiple […]
Cordis acquires drug-eluting balloon maker MedAlliance for up to $1.135B
MedAlliance announced today that Cordis acquired it for a total consideration that could reach up to $1.135 billion. The companies initially announced the planned deal in October of last year. Cordis, which develops interventional cardiovascular and endovascular technologies, made a $35 million investment last year. Its upfront closing payment totals $200 million, with achievement milestones […]
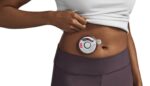
FDA approves wearable injector from Enable Injections
Enable Injections announced today that it received FDA approval for its enFuse injector for the delivery of Empaveli. Empaveli, commercialized by Apellis Pharmaceuticals, treats adults with paroxysmal nocturnal hemoglobinuria (PNH). The injector — in this case, called the Empaveli Injector — offers self-administration of the pharmaceutical. The compact, wearable device streamlines the self-administration experience with […]
Ypsomed has a new CFO, board member
Ypsomed announced today that it selected Samuel Künzli to take over the company’s chief financial officer (CFO) position. Niklaus Ramseier elected to hand over the reins to the CFO position after more than 20 years in the post. The transition begins on March 31, 2024, with Ramseier remaining available to the drug delivery device maker […]
Abbott: Real-world data indicates GLP-1s could be accelerator for FreeStyle Libre
New real-world data from Abbott (NYSE:ABT) demonstrates a potentially positive impact of GLP-1 drugs on diabetes technology. GLP-1 receptor agonists, like Ozempic and Wegovy, provide therapy for diabetes and weight loss. This therapeutic class, a glucagon-like peptide 1, has proven to lead to improved blood sugar control and weight loss. The drug class continues to […]
Satio wins $3.5M contract to develop at-home transdermal drug delivery device
Satio announced that a National Institutes for Health agency awarded it a $3.5 million Small Business Innovation Research (SBIR) contract. Awarded by the NIH’s Advanced Research Projects Agency for Health (ARPA-H), the contract helps the company develop SatioRx. This drug delivery device, compact and inexpensive, features disposable microneedle components for precise delivery. It remotely enables […]
Know Labs prices $7M public offering
Know Labs (NYSE:KNW) announced today that it priced an underwritten public offering worth about $7 million in proceeds. Seattle-based Know Labs, which develops a non-invasive glucose monitor, is offering 28 million shares of common stock. It priced the offering at 25¢ per share, with aggregate gross proceeds expected to total $7 million. The company expects […]
Senseonics completes 365-day CGM study cohort
Senseonics (NYSE:SENS) announced today that it completed its ENHANCE pivotal clinical study adult cohort for its 365-day Eversense CGM. Germantown, Maryland-based Senseonics’ ENHANCE study recorded the completed 365-day visit for the final study patient. The study evaluates the year-long implantable continuous glucose monitor in adults. It also has a pediatric cohort (ages 14 to 18), […]