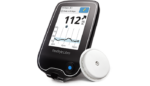
Dexcom (NSDQ:DXCM) topped expectations on Wall Street this week with its fourth quarter and full-year financial results. The San Diego, Calif.-based company posted a net loss of -$179.7 million, or -$2.03 per share, on sales of $338 million for the 3 months ended Dec. 31, for sales growth of 52.9% compared with the same period last […]
Featured
Abbott touts real-world data for FreeStyle Libre glucose monitor
Abbott (NYSE:ABT) presented real-world data this week showing that use of its FreeStyle Libre glucose monitoring system reduced prolonged hypoglycemia and helped users achieve better glucose control. At the 12th Advanced Technologies & Treatments for Diabetes meeting in Berlin, the company showcased a real-world evidence data set that included roughly 500,000 FreeStyle Libre system users. Pulling […]
Mylan launches generic sublingual film for opioid dependence
Mylan (NSDQ:MYL) said today that it launched its buprenorphine and naloxone sublingual film as a generic of Indivior‘s (LON:INDV) Suboxone sublingual film. The product is indicated for the treatment of opioid dependence. The therapy should be used alongside counseling and psychosocial support for people living with opioid dependence, according to Mylan. Sales for buprenorphine and naloxone […]
Propeller Health study suggests 84% of asthma inhaler use performed incorrectly
ResMed (NYSE:RMD) subsidiary Propeller Health today released results from a new study exploring real-world use of asthma rescue and controller inhalers that indicated that the majority of users did not use their inhalers properly. Results from the research were published in The Journal of Allergy and Clinical Immunology: In Practice, the Madison, Wisc.-based company said. Data in the study […]
ARS Pharmaceuticals wins fast track status for epinephrine nasal spray
ARS Pharmaceuticals said this week that it won fast track status from the FDA for its investigational intranasal epinephrine spray, ARS-1. The San Diego, Calif.-based company’s device is designed to be an easy-to-use treatment for people suffering from severe allergic reactions to food, drugs and insect bites. ARS Pharmaceuticals reported that the ARS-1 aqueous intranasal […]
Late mortality rates ‘inadvertently reversed’ in 5-year study of paclitaxel-eluting stent
The journal Circulation said this week that the all-cause mortality rates in a study comparing a paclitaxel stent and percutaneous transluminal angioplasty in people with peripheral artery disease were “inadvertently reversed” by the authors. In the first version of the paper, originally published in 2016, researchers reported that the 5-year all-cause mortality rate for people […]
Phillips-Medisize is offering connected health data services: Here’s how
One of the world’s largest medical device contract manufacturers, Phillips-Medisize is making a business play to provide the technological back end for connected drug delivery devices. Phillips-Medisize used to be all about the housings, assembly and packaging that went into finished drug-delivery devices. Electronics was something that other contract manufacturers did. Within a few years, […]
Abbott, Novo Nordisk ink deal for digital diabetes tools
Abbott (NYSE:ABT) said today that it inked a non-exclusive deal to integrate insulin dose data from Novo Nordisk‘s (NYSE:NVO) pre-filled and connected pens with its FreeStyle Libre mobile app and cloud-based system. The companies touted that integrating the products will allow healthcare professionals, caregivers and users to easily view glucose and insulin data, as well as […]
Ocular Therapeutix doses first wet-AMD patient in implant trial
Ocular Therapeutix (NSDQ:OCUL) said today that it dosed the first patient in a study of its OTX-TKI tyrosine kinase inhibitor implant. The Phase I trial is slated to assess the bioresorbable device’s safety, durability and tolerability in people with wet age-related macular degeneration. The company’s investigational product is a bioresorbable hydrogel fiber implant formulated with a […]
Hygieia wins FDA clearance for insulin management app
Hygieia said today that it won 510(k) clearance from the FDA for its insulin management app designed to optimize insulin dosing among people with Type 2 diabetes. The Michigan-based company touted its product as the first insulin management mobile app that can titrate individualized doses for all types of insulin regimens. It’s also the first […]