EyePoint Pharmaceuticals (NSDQ:EYPT) said today that it received $28.9 million in proceeds from the exercise of warrants by EW Healthcare Partners, Rosalind Advisors and another accredited investor. Last month, the Watertown, Mass.-based company won transitional pass-through status and reimbursement of its dexamethasone intraocular suspension, Dexycu, from the Centers for Medicare and Medicaid Services. The FDA-approved product […]
Featured
Fresenius cleared to jilt Akorn, court rules
The Delaware Chancery Court ruled this week that it can’t force Fresenius (ETR:FRE) to follow through with its $4.75 billion merger with Akorn (NSDQ:AKRX). The deal was designed to help Fresenius move into new dosage forms and therapeutic areas, including ophthalmic drugs, ear drops, nasal sprays and respiratory drugs. But Fresenius walked away from the deal in April, […]
Europe OKs Novo Nordisk’s Fiasp for Insulet’s Omnipod
Insulet (NSDQ:PODD) said today that its Omnipod insulin management system is available in Europe for use with Novo Nordisk‘s (NYSE:NVO) Fiasp fast-acting insulin aspart. Novo Nordisk’s Fiasp product enters the user’s bloodstream two times faster than NovoRapid and is designed to match a healthy body’s response to a meal, according to Insulet. “The addition of Fiasp for use […]
Johnson & Johnson closes $2B LifeScan sale
Johnson & Johnson (NYSE:JNJ) said today that closed the $2.1 billion sale of its LifeScan blood glucose monitoring subsidiary to private equity player Platinum Equity. The sale is part of the New Brunswick, N.J.-based healthcare conglomerate’s move to get out of the device end of the diabetes business. Early last year J&J announced a strategic review of the LifeScan, Animas […]
Ocugen kicks off dry eye disease trial for nanoemulsion eye drop
Ocugen said today that it launched a Phase III trial designed to evaluate its twice-daily 0.2% brimonidine tartrate eye drop in patients with dry eye disease. Ocugen plans to run two identical Phase III trials of OCU310, enrolling 240 participants per study. The company said it will assess safety and efficacy results after just 28 […]
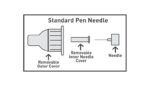
FDA counsels on proper insulin injection technique
The FDA issued a safety communication last week detailing the difference between standard pen needles and safety pen needles, both of which are used to inject medicine via pen injectors. A standard pen needle usually has an outer cover and a removable inner needle cover – both covers must be removed prior to an injection. […]
Abbott’s next-gen FreeStyle Libre wins CE Mark
Abbott (NYSE:ABT) said today that its next-generation FreeStyle Libre system won CE Mark clearance in the European Union. The glucose monitor, which eliminates the need for finger sticks, features optional alarms that users can customize, according to Abbott. “The FreeStyle Libre 2 system is designed to further empower people with diabetes by providing a choice about […]
J&J inks collab deal to develop MERS vaccine
Healthcare giant Johnson & Johnson (NYSE:JNJ) today announced a new collaborative deal as it looks to develop a vaccine against Middle East Respiratory Syndrome Coronavirus. The collaborative deal is between J&J’s Janssen Vaccines & Prevention division, the Coalition for Epidemic Preparedness Innovations and The Jenner Institute at the University of Oxford, J&J said. Through the deal, Janssen […]
Gerresheimer’s Sensile Medical wins CE Mark for infusion pump
Gerresheimer subsidiary Sensile Medical said today it won CE Mark approval in the European Union for its wearable micropump intended for use in the treatment of Parkinson’s disease. The newly cleared device features automatic filling for use with liquid medicines, and includes a color display, charging unit and leather bag which allows the device to be worn […]
Valeritas kicks off device registration process in China
Valeritas (NSDQ:VLRX) said today that it began the process to register its wearable insulin delivery device, V-Go, in China. The company noted that it signed an agreement with an unnamed contract research organization to work directly with the China National Drug Administration, in the hopes of winning regulatory approval for V-Go in China. “The China market […]