Abbott (NYSE:ABT) is slated to pay $25 million upfront for the global commercialization rights to Surmodics‘ (NSDQ:SRDX) SurVeil drug-coated balloon, the companies announced today. The company’s drug-device combo, which is in pivotal trials in the U.S., is designed to treat peripheral artery disease in the superficial femoral artery. According to the deal, Abbott could also negotiate […]
Featured
Medtronic thinks these clinics could be the future of diabetes care – here’s why
Updated to include comments from David Kaplan. Managing a chronic illness can feel like a full-time job. Between juggling multiple appointments with an array of specialists and following a meticulous care plan, patients dealing with conditions like diabetes have a lot on their plate. Medtech titan Medtronic (NYSE:MDT) wants to help change that. The company’s CEO […]
Anthem exec chairman joins Proteus board | Personnel Moves – Feb. 26, 2018
Proteus Digital Health announced earlier this month that Anthem‘s (NYSE:ANTM) executive chairman, Joseph Swedish, joined the company’s board of directors. Swedish served as Anthem’s president & chief executive from 2013 until he was appointed executive chairman in 2018. “Proteus has created a compelling solution to a problem that has plagued the pharmaceutical industry for decades: […]
Lilly touts Ph3 data for once-weekly Type II diabetes medication
Eli Lilly (NYSE:LLY) touted data today from a Phase IIIb study of its once-weekly, injectable Type II diabetes medication, Trulicity. The insulin-maker reported that its GLP-1 receptor agonist significantly improved blood sugar control in people with Type II diabetes when added to ongoing treatment with a SGLT-2 inhibitor. “I’ve seen in my practice that even with […]
Patent board rules Mohawk tribe is not immune to challenges over Restasis IP
The U.S. Patent Trial and Appeal Board ruled last week that the tribal immunity granted to Native American tribes in the U.S. does not shield tribes from patent challenges. The ruling is a setback for Allergan (NYSE:AGN); the pharmaceutical company transferred patent rights for its blockbuster eye drug, Restasis, to the St. Regis Mohawk Tribe last […]
Medtronic wins FDA nod for smallest drug-eluting stent on U.S. market
Medtronic‘s (NYSE:MDT) 2.0-mm Resolute Onyx drug-eluting stent has won FDA approval and is set to launch in the U.S., making it the smallest device of its kind on the market. The stent is designed to treat patients with coronary artery disease who have small vessels. Interventional cardiologists often face challenges treating this patient group with […]
Reva Medical wins CE Mark for bioresorbable Fantom Encore scaffold
Reva Medical (ASX:RVA) has won CE Mark clearance for its thin strut Fantom Encore bioresorbable, drug-eluting scaffold. The sirolimus-eluting vascular scaffold, which is made from Reva’s Tyrocore polymer, features a 95-micron strut profile and x-ray visibility. “Fantom Encore has the thinnest strut profile of any available bioresorbable scaffold in the 2.5 millimeter diameter size. A thinner […]
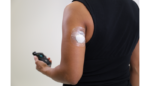
Medtronic wins sensor nod from FDA for MiniMed 670G auto insulin tech
The FDA has cleared Medtronic‘s (NYSE:MDT) Guardian sensor, part of the medtech titan’s MiniMed 670G automated insulin delivery system, to be worn on the upper arm. The device is Medtronic’s newest and most accurate continuous glucose monitor, the company touted, and is the only sensor approved by the FDA for use with a hybrid-closed loop […]
KemPharm wins FDA nod for short-term pain prodrug
Shares in KemPharm (NSDQ:KMPH) soared today after the company said that the FDA approved a re-submitted application for its pain-relief prodrug, Apadaz. The product, which combines benzhydrocodone and acetaminophen, is indicated for the short-term management of acute pain. The FDA rejected KemPharm’s first attempt at regulatory approval for Apadaz in 2016. Today’s win for KemPharm is […]
Novo Nordisk touts promising results from late-stage trial of oral diabetes med
Setting the stage for a battle with rival Eli Lilly (NYSE:LLY), Novo Nordisk (NYSE:NVO) reported data this week from a late-stage trial of its oral formulation of semaglutide, a medication for people with Type II diabetes. The GLP-1 agonist is traditionally administered via injection. Both insulin-makers have approved versions of the injectable semaglutide, but Novo Nordisk is […]