Inovio Pharmaceuticals (NSDQ:INO) said today that the FDA placed a hold on its proposed Phase III clinical program for its investigational DNA immunotherapy candidate designed to treat pre-cancers and cancers caused by human papillomavirus. The California-based company completed a randomized, double-blind Phase II trial of the candidate in July 2014, evaluating the DNA immunotherapy treatment in women […]
Oncology
Bacterial nanostructures developed to locally deliver proteins
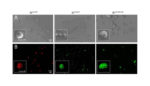
Spanish researchers demonstrated that bacterial inclusion bodies, non-toxic nanostructures, hold promise for a sustained, local delivery of protein drugs by mimicking components of a mammal’s endocrine system. New materials developed as controllable, localized systems of drug delivery often aim to be immobilized in a given tissue as a drug “depot” for long term supply of […]
Gold nanoparticles hold promise for pancreatic cancer treatment
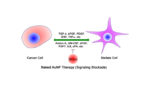
Gold nanoparticles could be an important part of developing a treatment for pancreatic cancer, according to new research published yesterday in the journal ACS Nano. Previous work demonstrated that gold nanoparticles can be an effective vehicle to carry chemotherapy drugs into tumors. They can also act as a target to enhance the impact of radiation on […]
Keystone Nano wins orphan designation for ceramide liver cancer treatment
Keystone Nano said today that its ceramide nanoliposome won orphan drug status from the FDA for the treatment of liver cancer. The designation provides the company with a window of market exclusivity after product approval. The State College, Penn.-based company touted its nanoliposome delivery system as being able to load both water-loving and water-hating compounds and […]
FDA awards product development grants for rare diseases
The FDA said yesterday that it awarded 21 clinical trial research grants totaling more than $23 million over the next 4 years to stimulate product development for patients with rare diseases. The Orphan Products Clinical Trials Grants Program has awarded more than $370 million to fund more than 590 clinical studies since its creation in […]
Tunable hydrogel helps predict tissue response to chemotherapy
Researchers from the Wyss Institute developed a hydrogel microenvironment to evaluate how physical properties like stiffness of the extracellular matrix impacts the efficacy of chemotherapy. “To have success with chemotherapy and other drug therapies, we will likely need to screen their effectiveness against cells living in various environments, and not just assume that cells will always […]
Novocure enrolls 1st patient in Phase III brain metastases trial
Novocure (NSDQ:NVCR) said that the 1st patient has been enrolled in a Phase III pivotal trial of its tumor-treating field technology in combination with radiosurgery to treat brain metastases from non-small-cell lung cancer. The St. Helier, N.J.-based company’s tumor treating fields (TTFields) are low-intensity, intermediate-frequency, alternating electric fields designed to disrupt cell division within cancer cells. Novocure’s commercial product, Optune, […]
‘Connectosomes’ efficiently deliver chemo to targeted cells
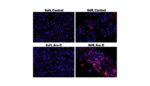
Researchers have developed a new form of nanoparticles called “connectosomes” that can efficiently deliver chemotherapy to human cells more efficiently than traditional delivery methods, according to a report out of the University of Texas at Austin. The team, who published their research in the Journal of the American Chemical Society, equipped the connectosomes with gap junctions […]
Motion-directed microrobots for targeted drug delivery
Researchers have created tiny ‘microbots’ that are able to move and react in a fashion similar to bacteria, according to a report from the Max Planck Institute for Intelligent Systems. Bacteria use flagella to move, directed by stimuli like sources of light or the earth’s magnetic field. Researchers have developed synthetic microrobots with motility and phototaxis, […]
Dicerna bails on EnCore lipid nanoparticle tumor delivery program
Dicerna Pharmaceuticals (NSDQ:DRNA) said yesterday that it plans to spike the development program for its lipid nanoparticle delivery system for solid tumors because its preliminary results don’t meet expectations. The Cambridge, Mass.-based company evaluated its lipid nanoparticle, DCR-MYC, in a pair of clinical trials – a Phase I study of patients with solid tumors, hematological […]