Delcath Systems (NSDQ:DCTH) won a lot of Wall Street love today on news that it landed the 1st commercial order for its organ-targeting chemotherapy system, the ChemoSat device.
DCTH shares spiked 12.% to $3.46 in morning trading today, a positive turn for the company, which has seen its stock take a rocky roll downhill since hitting $4.49 early in February. Delcath, however, is no stranger to the ups and down of the market.
"Delcath is an extremely volatile stock and there a number of components to that," CEO Eamonn Hobbs told MassDevice last summer. "One is that it’s a stock that’s shareholder based, that’s dominated by retail shareholders who tend to be much more active traders. The trading volume is high and the volatility seems to be equally high. The stock seems to go up significantly on good news and down significantly on bad news — or no news."
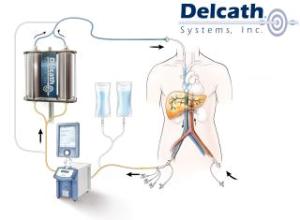
The European Institute of Oncology became the 1st owner of a commercial ChemoSat system, which uses a system of tubes to localize the delivery of toxic chemotherapy agents by isolating the targeted organ from the rest of the body’s bloodstream.
The New York-based device maker has been padding its nest in Europe with the recent announcement of new training centers in France and Spain, and the company said it’s picking up the pace in hiring and training its direct sales teams.
"The first commercial order of our ChemoSat system is a significant milestone in Delcath’s history, marking our transition from a research and development organization to a commercial enterprise," Hobbs said in prepared remarks. "We believe that our momentum is building and we are taking critical steps toward our goal of establishing CHEMOSAT as a new treatment option for patients with cancers in the liver in Europe."
Check out our interview with Delcath president & CEO Eamonn Hobbs from June, 2011.
Delcath has had a rough go with the ChemoSat system in the last year with inconclusive trial results and a 5 million share stock offering that didn’t win much favor from The Street.
The troubles began when the FDA refused the company’s new drug application over concerns that the information provided was insufficient for review. ChemoSat is regulated as a medical device in Europe, where it won CE Mark approval in April 2011, but it is regulated as drug-device combination that also requires new drug labeling as far as the FDA is concerned.
"The CE Mark that we have in Europe is a very important regulatory approval, because virtually every country in the world outside of the U.S. accepts the CE Mark as a predicate regulatory approval and provides reciprocal approval," Hobbs told us in an exclusive interview in June 2011. "We’ll be leveraging the CE Mark around the world to gain regulatory approval in all the emerging markets in relatively short order. We expect to have regulatory approval in countries in Asia, South America, Central America and Canada, the Middle East and Africa starting in 2012. We should have regulatory approval in virtually all the countries by the end of 2013."
The company touted a regulatory win in Australia last month.

