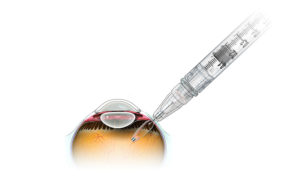
The PDS is a permanent, refillable eye implant that’s the approximate size of a grain of rice. It offers continuous delivery of a customized formulation of ranibizumab over a period of months to reduce the treatment burden associated with frequent eye injections.
South San Francisco, Calif.–based Genentech, a member of the Roche Group, said in a news release that the FDA accepted the BLA under priority review to treat wet or neurovascular age-related macular degeneration (AMD).
The company said that, if approved, PDS would offer a first-of-its-kind therapeutic approach to offer those with wet AMD an alternative to frequent eye injections of anti-vascular endothelial growth factor (VEGF), the current standard of care. Genentech expects an FDA decision on approval by Oct. 23, 2021.
BLA submission follows positive results from Genentech’s Phase 3 Archway study primary analysis that demonstrated that, of wet AMD patients treated with PDS, more than 98% could go six months without needing additional treatment before the refill exchange.
Patients in the trial also achieved vision outcomes equivalent to patients receiving monthly ranibizumab eye injections, while PDS was generally well-tolerated with a favorable benefit-risk profile. If approved, PDS would be the first and only wet AMD therapy indicated to allow six months between treatments, Genentech said.
A PDS marketing authorization application also received validation from the European Medicines Agency and is currently under review.
“Anti-VEGF therapy brings significant benefit to people with wet AMD, but optimal results require frequent trips to the doctor’s office for eye injections. This burden leaves many people under-treated and susceptible to vision loss,” Genentech CMO & head of global product development Dr. Levi Garraway said in the release. “If approved, PDS would transform wet AMD treatment by providing up to six months of uninterrupted therapy that could potentially improve vision outcomes compared to what is currently achieved in the clinic.”

