The FDA agreed to review Delcath Systems’ (NSDQ:DCTH) new drug application, bringing the device maker 1 step closer to launching its organ-isolating chemotherapy system on the U.S. market.
The federal watchdog agency designated the application, Delcath’s 2nd attempt at approval, for standard review. Delcath expects to be notified of its panel date by the end of the month, according to a press release.
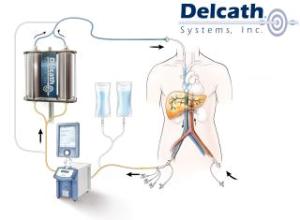
Delcath’s ChemoSat uses a system of tubes to localize the delivery of toxic chemotherapy agents by isolating the targeted organ — in this case, the liver — from the rest of the body’s bloodstream.
Delcath must seek new drug approval for the melphalan hydrochloride the ChemoSat system uses to destroy cancerous tissue.
The device maker submitted its 2nd new drug application in August after the FDA refused the original NDA, filed last year, over concerns that the information provided was insufficient for review.
Check out our interview with Delcath president & CEO Eamonn Hobbs.
"FDA acceptance of our NDA is a significant milestone for the company, and we look forward to working closely with the agency throughout the review process with the goal of securing approval of our application," president & CEO Eamonn Hobbs said in prepared remarks. "We believe that the standard review period will provide both Delcath and the FDA a manageable time-frame to thoroughly review the combination product submission. Our most important objective is to be able to provide patients with unresectable metastatic melanoma in the liver a new option for treating their disease."
DCTH shares gained 1.2% on the news yesterday, closing at $1.72.

