 Researchers from the Massachusetts Institute of Technology and Brigham & Women’s Hospital have developed a novel, responsive drug-delivery material that can reside in the stomach for up to nine days.
Researchers from the Massachusetts Institute of Technology and Brigham & Women’s Hospital have developed a novel, responsive drug-delivery material that can reside in the stomach for up to nine days.
The teams work was published today in Nature Communications.
“One of the biggest issues in health care is noncompliance, people simply not taking their drugs,” co-senior author Robert Langer said in prepared remarks. “We have been working with the Bill and Melinda Gates Foundation to develop ultra-long-lasting capsules, which might last for the entire course of a treatment, or could be taken once a week or once a month, depending on the device.”
An ingestible material that is capable of residing in the GI tract has to be designed with a number of factors in mind – an ingestible capsule has to be small enough for a patient to swallow, but large enough that it won’t be passed out of the stomach.
The team of researchers also wanted to build in a trigger that would cause the device to self-destruct, in case a patient experienced an allergic reaction or an adverse side effect to the drug-device combination.
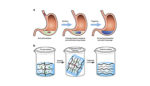
Traditionally, hydrogels have been seen as a useful material in ingestible drug-delivery applications – a patient could swallow a dehydrated hydrogel capsule that would swell once inside the stomach.
But hydrogels are soft and don’t do well in the stomach’s compressive forces, according to the researchers from MIT and Brigham & Women’s Hospital. Instead, the team integrated two polymer networks to create a tougher material.
“There are two networks. One is composed of alginate, a material derived from seaweed, and the other is polyacrylamide, a widely-used polymer,” co-senior author Giovanni Traverso said.
To crosslink the networks, the team used two types of chemical bonds, which can each be triggered to dissolve using glutathione and EDTA. If the drug-delivery device needed to be removed from the stomach quickly, the patient could just swallow these trigger compounds, the team reported.
Upon testing the materials, the researchers observed that they were strong enough to resist fracture. They also tested devices made from the tough material in a pig model and found that it was able to withstand the stomach’s harsh environment for more than seven days.
The team also tested the device as a drug delivery system. They found that it was able to release an antimalarial drug, lumefantrine, over the course of several days.
Next, the researchers plan to characterize the rate of drug-release from capsules made from the crosslinked polymer network, as well as pursue other applications for the material.

