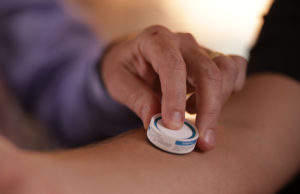
Cambridge, Massachusetts-based Vaxxas picked up AU$8.2 million ($5.48 million). The second-round grant represents part of the Australian government’s Modern Manufacturing Initiative (MMI). Funding supports the manufacturing scale-up of Vaxxas’ device for applying vaccines to the skin using a small patch.
The company received AU$4.4 million (nearly $3 million) in a first-round MMI in 2021. That grant supported the specialized infrastructure needed to manufacture the high-density microarray patch (HD-MAP). Manufacturing took place at the company’s currently under construction Northshore, Brisbane, Australia facility.
Additional support from the Australian government aims to help Vaxxas develop and establish a sterile production line. The line uses proprietary aseptic fill and finish processes at the facility. The goal is to advance the company’s vaccine coating, device sealing and quality control technologies. The Brisbane facility also exists to support late-stage clinical trials and early commercial production.
“We are incredibly grateful to the Australian federal government for their ongoing support of our vaccination technology,” Vaxxas CEO David Hoey said in a news release. “It is fantastic to see the government backing innovations by local companies that have the potential to make a difference to lives in Australia and around the world.”
The Vaxxas Brisbane facility is slated to open in early 2023. The existing (relocated) workforce of more than 100 employees is set to add 29 new highly skilled jobs. Vaxxas plans to work with the Department of Industry, Science and Resources over the coming weeks to finalize grant details.
About the Vaxxas technology
Vaxxas designed its HD-MAP technology to use an ultra-high-density array of micro projections, invisible to the human eye. Applied to the skin as a patch, the system rapidly delivers vaccines to the abundant immune cells immediately below the skin surface.
The company said its technology could potentially improve pandemic and routine vaccination efforts. It offers production efficiencies, breadth and speed of access, plus population coverage.
In October 2020, Vaxxas received a $22 million award from the Biomedical Advanced Research and Development Authority (BARDA) at the U.S. Dept. of Health and Human Services (HHS) to support a clinical study for the patch.
The company licensed a COVID-19 vaccine from The University of Texas at Austin earlier this year. It aims to create the first needle-free, room-temperature, stable COVID-19 vaccine patch to enter clinical studies.

