Robotic drug delivery technology developer Bionaut Labs announced today that it closed a Series B financing round worth $43.2 million. Los Angeles-based Bionaut Labs uses microscale robots to deliver drugs for treating central nervous system (CNS) diseases and disorders. Through magnetic propulsion, the company’s Bionauts can navigate the human body and deliver drugs locally. The […]
Oncology
TriSalus releases positive data for its catheter-based drug delivery tech
TriSalus Life Sciences today announced positive clinical results for its Pressure-Enabled Regional Immuno-Oncology technology. The company, currently in the process of going public through a SPAC merger, filed a presentation for investors. It features information regarding its ongoing PERIO 01 and PERIO 02 clinical studies. The studies evaluate the TriNav pressure-enabled drug delivery technology for […]
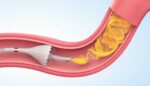
FDA approves Medtronic drug-eluting stents for treating bifurcation lesions
Medtronic (NYSE:MDT) announced today that the FDA approved its Onyx drug-eluting stents (DES) for treating certain lesions. Approval covers the treatment of non-left main bifurcation lesions utilizing the provisional bifurcation stenting technique. The technique uses a single stent to treat the bifurcation in percutaneous coronary interventions (PCIs). It applies to the FDA-approved and CE-marked Onyx […]
FDA clears embolic microspheres from ABK Biomedical
ABK Biomedical announced today that it received FDA 510(k) clearance for its Easi-Vue embolic microspheres for treating tumors. Halifax, Nova Scotia-based ABK designed its microspheres for treating patients with arteriovenous malformations and hypervascular tumors. Vascular embolization is an effective therapy for addressing symptoms of these conditions. ABK said in a news release that microsphere embolization […]
Study backs Fluidx embolic device for vessel filling
Fluidx Medical announced today that study results support vessel filling with its GPX embolic device when compared to microspheres. Salt Lake City-based Fluidx designed the GPX embolic device for simple preparation and controllable material delivery, packaged ready-to-use in a syringe and capable of being prepped tableside by a clinician in about 30 seconds. It may […]
FDA approves first targeted infusion therapy for HER2-low breast cancer
The FDA announced today that it approved Enhertu, an IV infusion for the treatment of patients with unresectable or metastatic HER2-low breast cancer. Enhertu (fam-trastuzumab-deruxtecan-nxki) IV infusion is now the first approved therapy targeted to patients with the HER2-low breast cancer subtype, which is a newly defined subset of HER2-negative breast cancer. The therapy received […]
Inovio names new chief medical officer
Inovio (Nasdaq:INO) announced today that it appointed Dr. Michael Sumner as its new chief medical officer (CMO). Sumner will oversee Inovio’s clinical-stage pipeline of DNA medicines, global clinical development, clinical operations and biostatistics efforts, along with regulatory affairs, pharmacovigilance and medical affairs. He will also serve on the company’s executive leadership team and report to […]
Boston Scientific warns on some SpaceOAR systems
Boston Scientific (NYSE:BSX) has issued an urgent field safety notice in Europe due to potential issues with its SpaceOAR systems. Marlborough, Massachusetts-based Boston Scientific’s SpaceOAR system is an absorbable polyethylene glycol hydrogel spacer for separating the prostate from the rectal wall during radiation treatment for prostate cancer. The SpaceOAR Vue, the next-generation hydrogel spacer, offers enhanced […]
First patients enrolled in trial for Boston Scientific’s SpaceOAR Vue hydrogel
GenesisCare announced today that it enrolled the first patients in a trial for Boston Scientific’s (NYSE:BSX) SpaceOAR Vue hydrogel. Boston Scientific’s SpaceOAR Vue hydrogel will be evaluated in the SABRE trial, sponsored by Boston Scientific, for prostate cancer patients receiving stereotactic body radiation therapy (SBRT). Get the full story at our sister site, MassDevice.
Dyve Biosciences to collaborate with Moffitt Cancer Center on transdermal buffering agent
Dyve Biosciences announced today that it entered into a joint two-year research collaboration with the Moffit Cancer Center. Under the collaboration, Dyve and Moffit will study the former’s systemic buffering agent, DYV800, delivered through the company’s broadly applicable transdermal delivery technology, in various cancer laboratory models. According to a news release, the models through which […]