By: Theresa M. Mullin, Ph.D. Increasingly, drug development is a global endeavor. It requires international collaboration to ensure that consistent standards are adopted and adhered to by all drug makers and regulatory authorities, regardless of country of origin or destination. I am pleased to have been in China recently, when the China Food and Drug […]
This new nanochip technology can reprogram human cells
Ohio State University researchers have developed a nanochip technology that they say can create any cell type for treatment within the human body. The new technology, called Tissue Nanotransfection (TNT), can repair injured tissue and restore the function of aging tissue like organs, blood vessels and nerve cells. “By using our novel nanochip technology, injured […]
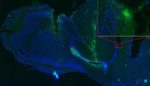
Electrical fields can heal brain damage: Here’s how
Electrical fields can guide neural stem cells into a specific location to repair brain damage, according to new research from the University of California at Davis. Min Zhao, a researcher at UC Davis, studies how electric fields can guide wound healing. His previous research has shown that electric fields are able to attract cells into […]
FDA unveils digital health technology changes
By Stewart Eisenhart, Emergo Group Medical device regulators at the US Food and Drug Administration’s Center for Devices and Radiological Health (CDRH) have published further details on their plans to clarify and improve policies regarding mobile medical applications, wearables and other digital health products. Get the full story here at the Emergo Group’s blog. The […]
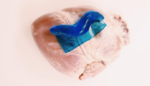
How slugs are creating better medical adhesives
Slug mucus is the inspiration behind a new adhesive to close surgical wounds and reduce the use of surgical staples, according to new research out of Harvard. Some of the current adhesives on the market can be toxic and stick together tissues weakly. Some can’t be used in wet environments altogether, which can pose a […]
Medtech stories we missed this week: August 4, 2017
From MDCorp inking a European distribution deal to Align Technology’s North Carolina expansion, here are seven medtech stories we missed this week but thought were still worth mentioning. 1. MDCorp inks distribution deal with Smart Endoscope Systems MDCorp announced in a July 31 press release that it has signed a distribution agreement with Smart Endoscope […]
Chinese regulators answer two common clinical trial questions
By Stewart Eisenhart, Emergo Group China Food and Drug Administration (CFDA) regulators have addressed frequently asked questions regarding clinical trial requirements for medical devices. Get the full story here at the Emergo Group’s blog. The opinions expressed in this blog post are the author’s only and do not necessarily reflect those of DrugDeliveryBusiness.com or its […]
This tiny diaphragm pump could enable medical device innovation
Fraunhofer researchers have created a tiny yet powerful diaphragm pump that can deliver ambient air to gas sensors. The sensors are attached to a smartphone that warns a user of heavy exposure to particulate matter. “Our smart pump measures only 25 sq mm, making it the world’s smallest pump. That said, it still has a […]
Using robot assistance in neurosurgery for faster seizure mapping
Head shaved, a little boy rests on the operating table, deep under anesthesia. His parents have brought him to Boston Children’s Hospital in hopes of determining the cause of his seizures. Now, neurosurgeons Scellig Stone, MD, PhD, Joseph Madsen, MD, and their colleagues in the Epilepsy Center are performing a procedure designed to monitor seizure […]
India publishes essential principles for medical device and IVD safety and performance
By Stewart Eisenhart, Emergo Group The Central Drugs Standard Control Organization (CDSCO), India’s medical device market regulator, is seeking input from industry on draft essential safety and performance principles included in new regulations launching in 2018. Get the full story here at the Emergo Group’s blog. The opinions expressed in this blog post are the […]