By: Scott Gottlieb, M.D. It is incumbent upon FDA to ensure that we have the right policies in place to promote and encourage safe and effective innovation that can benefit consumers, and adopt regulatory approaches to enable the efficient development of these technologies. By taking an efficient, risk-based approach to our regulation, FDA can promote […]
Peruvian regulators push for faster medical device approval timeframes
By Stewart Eisenhart, Emergo Group The Peruvian government has rolled out a raft of updates to the country’s medical device regulatory system, including an initiative to reduce approval timeframes for all device classes. Get the full story here at the Emergo Group’s blog. The opinions expressed in this blog post are the author’s only and […]
FDA expands 510(k) validation data requirements for reusable devices
By Stewart Eisenhart, Emergo Group Medical device market regulators at the Food and Drug Administration have issued a new list of reusable medical devices for which validated Instructions for Use as well as validation data for cleaning and sterilization will be required for 510(k) submissions. Get the full story here at the Emergo Group’s blog. […]
How creative FDA regulation led to first-in-the-world approval of a cutting-edge heart valve
By: Jeffrey Shuren, M.D., J.D., and Bram Zuckerman, M.D. Nearly six years ago FDA approved an artificial transcatheter heart valve (THV) to treat patients having severe symptoms and life-threatening heart problems such as fainting, chest pain, heart failure, irregular heart rhythms, or cardiac arrest, because one of the valves in their heart (the aortic valve) […]
South African government forms medical device and drug market regulatory body
By Stewart Eisenhart, Emergo Group The South African government has formed a new agency to oversee new medical device and pharmaceutical regulatory systems being developed in the country. Get the full story here at the Emergo Group’s blog. The opinions expressed in this blog post are the author’s only and do not necessarily reflect those […]
Statement from FDA Commissioner on opioid drugs with abuse-deterrent properties
Last month, I asked my colleagues at the FDA to identify what additional and more forceful steps the FDA can take, on top of the vigorous work the agency is already doing, to address the crisis of opioid addiction. Everyone at the FDA is committed to focusing on all aspects of the epidemic. The new […]
Battery-free pacemaker successfully powers the heart
A wireless, battery-free pacemaker has shown success in powering the heart in pig models, according to researchers from Rice University and Texas Heart Institute. The pacemaker, designed by lead researcher Aydin Babakhani, gets its energy wirelessly from radio frequency radiation that gets transmitted wirelessly from an external battery pack from a few centimeters away. Get […]
This microhole chip identifies and sorts cancer cells
Fraunhofer researchers have created a microhole chip that can identify and characterize cancer cells within minutes – helping to catch metastasis before it can begin. Traditional fluorescence-activated cell sorting (FACS) gives an estimate of the number of tumor cells in a patient’s bloodstream. If there is a higher concentration of tumor cells, there is a […]
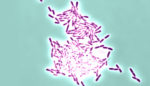
This bacterial discovery could prevent biofilms
Researchers at the University of Texas at Austin have discovered a potential new way to eliminate biofilms to reduce the number of hospital-borne infections. Biofilms are a group of microorganisms that stick to a surface. Over time, the cells in the biofilm can grow and cause chronic infections and can even become resistant to antibiotics. […]
Diba Industries appoints new president
Diba Industries (Danbury, Conn.) has announced that Timothy O’Sullivan has been appointed as the company’s president, effective June 1. O’Sullivan’s transition to company president should come easy, after having served at the president of Diba Industries’ Bio-Chem Fluidics for 6 years. He led significant business changes at Bio-Chem Fluidics like reinvigorating the vision and culture of […]