Children’s National Health System touted data today from a Phase I dose-ranging study evaluating a cell therapy approach in patients with blood and bone marrow cancers for whom stem cell transplantation hasn’t worked. Results from the Resolve trial showed that 78% of patients responded to multi-tumor-associated antigen specific lymphocytes (TAA-L) treatment and 44% of patients […]
GlaxoSmithKline, Innoviva tout data for Relvar Ellipta inhaled drug combo
GlaxoSmithKline (NYSE:GSK) and Innoviva (NSDQ:INVA) touted data today from a non-inferiority lung function study evaluating its inhaled, once-daily corticosteroid/long-acting beta2 agonist combination therapy in patients with asthma. The study showed that patients with well-controlled asthma could switch from the twice-daily Seretide Accuhaler to the companies’ once-daily Relvar Ellipta therapy without compromising lung function. Patients who switched to […]
Amgen wins marketing authorization for Repatha single-dose delivery option
Amgen (NSDQ:AMGN) said today that the European Commission granted a change to the marketing authorization of its Repatha device, approving a single-dose delivery option. The pre-filled cartridge mini-doser is a hands-free device that is designed to provide 420 mg of Repatha (evolocumab) in a single injection for patients with cardiovascular disease. The company touted its […]
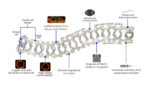
Meril’s bioresorbable, drug-eluting vascular scaffold
Meril Life Sciences has developed a drug-eluting, bioresorbable vascular scaffold that the body can slowly degrade and wash away. The company’s MeRes100 is made of biodegradable polymers, PLLA and PDLLA. Sirolimus, a commonly-used immunosuppressant found in drug eluting stents, is embedded in the scaffold’s coating. The company designed the struts of the device to be just 100 μm […]
BrainStorm seeks early Health Canada nod for NurOwn treatment
Brainstorm Cell Therapeutics (NSDQ:BCLI) is reportedly seeking early regulatory approval in Canada for its NurOwn stem cell treatment for patients with ALS. The company told Reuters this week that it has inked an agreement with a Canadian non-profit organization, CCRM, that helps to support the development of regenerative medicine. The organization plans to help BrainStorm meet Canadian regulatory requirements […]
Alimera, Knight seek Canadian regulatory nod for Iluvien intravitreal implant
Alimera Sciences (NSDQ:ALIM) and Knight Therapeutics (TSE:GUD) said today that Health Canada accepted the companies’ New Drug Application for its intravitreal implant, Iluvien. The FDA approved Atlanta-based Alimera’s sustained release intravitreal implant in 2014 to treat diabetic macular edema in patients who have been previously treated with corticosteroids and did not have a clinically significant rise in […]
FDA accepts Ocular’s NDA resubmission for Dextenza
Ocular Therapeutix (NSDQ:OCUL) said today that the FDA accepted the company’s resubmitted New Drug Application for its post-surgical ocular pain reliever, Dextenza. The hydrogel plug, inserted into a patient’s tear duct, is designed to deliver a sustained dose of dexamethasone over 4 weeks following opthalmic surgery. The Bedford, Mass.-based company has had trouble gaining regulatory approval […]
Study: Industry funding biases drug trials in favor of sponsors
Researchers from the University of Sydney published a review this week, concluding that industry-funded drug trials are often biased in favor of the sponsors’ products. According to Lisa Bero of the university’s Charles Perkins Centre, the study provides “definitive evidence that pharmaceutical industry funding of drug studies biases the results and conclusions to look favourable […]
Researchers develop smart drugs using ‘DNA computers’ for controlled delivery
Researchers from the Eindhoven University of Technology have developed the 1st “DNA computer” that can detect several antibodies in the blood and perform calculations based on what it detects. The team published their work in Nature Communications and said that it’s an important step towards developing smart drugs that allow for more targeted and controlled medication delivery. […]
Common Sensing, Welkin Health collaborate on digital diabetes management system
Common Sensing and Welkin Health said last month that the companies are partnering to develop a digital diabetes management program designed to improve insulin adherence. The companies plan to craft a program that combines insulin dosing data collected from Common Sensing’s Gocap smart insulin pen with Welkin Health’s digital coaches to support patients with diabetes. […]