Abbott (NYSE:ABT) divested 60% of its remaining stake in Mylan (NSDQ:MYL) this week, according to regulatory filings, disposing of 44 million shares at $41.60 apiece. The trade was valued at $1.7 billion. In 2014, Mylan bought Abbott’s generics business outside the U.S. in a deal that rang in at nearly $5.3 billion. The deal allowed Canonsburg, Penn.-based Mylan […]
Gamida Cell wins additional EMA orphan drug designation for NiCord
Gamida Cell said today that the European Medicines Agency granted its NiCord cell graft an additional orphan drug designation as a treatment for hematopoietic stem cell transplantation, or bone marrow transplantation. The treatment is a graft derived from umbilical cord blood which has been expanded and enriched with stem and progenitor cells, according to the […]
BioClin Therapeutics raises $30m to advance antibody-drug combo therapy
BioClin Therapeutics said yesterday that it closed a $30 million Series B round led by new investors Sofinnova Ventures and Ysios Capital. Existing investors HealthCap, Life Sciences Partners and Tekla Capital Management also invested. The company plans to use the newly-acquired funds to advance its lead candidate, B-701, as a treatment for metastatic bladder cancer in […]
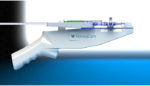
Intact Vascular finishes patient enrollment for angioplasty clinical trial
Intact Vascular said today that it finished enrollment for the Toba II clinical trial evaluating its Tack Endovascular System in combination with standard or drug coated balloon angioplasty in arteries above the knee. The company’s trial enrolled 210 patients and will focus on the superficial femoral artery and the proximal segment of the popliteal artery, […]
Tandem Diabetes Care prices $22.5m public offering
Tandem Diabetes Care (NSDQ:TNDM) today announced the pricing details of a $22.5 million underwritten public offering. The company priced 18 million shares of its common stock at $1.25 apiece. Tandem also gave underwriters a 30-day option to buy up to an additional 2.7 million shares of common stock at the public offering price. The offering is expected […]
The stem cell ‘skin gun’ that’s aiming to disrupt wound care
RenovaCare’s SkinGun uses a patient’s own stem cells to heal their wounds faster and more efficiently than a traditional skin graft. Pennsylvania state police officer Matthew Uram suffered severe second-degree burns to his face, right arm and leg after a friend’s bonfire got out of control. Uram was facing months of painful skin grafts, the […]
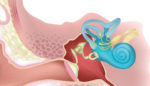
The stem cell therapy that could reverse hearing loss
Frequency Therapeutics is betting that it can change the lives of millions of Americans with hearing loss, by triggering the body’s natural ability to heal itself. Researchers have used cells in regenerative medicine for decades – in 1931, the father of cell therapy, Paul Niehans, treated a patient with material from calf embryos. Although today’s […]
Mylan recalls 81,000 EpiPens over reports of device failure
Mylan (NSDQ:MYL) recalled 81,000 EpiPen emergency allergy auto-injectors this week after receiving 2 reports that the device failed to work in emergencies. The recall affects devices in Australia, New Zealand, Europe and Japan, the company reported. Canonsburg, Penn.-based Mylan said that in both cases, patients received treatment using an alternate EpiPen. The drugmaker also said that it […]
Press Release: Nemera wins the “Editor’s choice” Exhibitor Award at Interphex with its autoinjector Safelia
(Press release) – International Pharmaceutical Expo (INTERPHEX), the premier event dedicated to pharmaceutical and biotechnology innovation, technology, and knowledge from development through commercialization and sponsored by the Parenteral Drug Association (PDA), has announced the winners for the INTERPHEX Exhibitor Awards for 2017. Nemera was recognized for the ‘Editor’s choice’ for Safelia. Safelia is an innovative […]
Origin treats first patients in study of nitric oxide for diabetic foot ulcers
Origin said today that it treated the 1st patients in its dose-ranging Genesis Phase IIb-equivalent trial. The study is evaluating therapeutic quantities of plasma-generated nitric oxide as a treatment for chronic diabetic foot ulcers. The 27-week trial is slated to enroll up to 100 patients. Patients will be randomized into 1 of 4 dosing regimens, […]