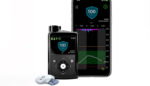
Medtronic (NYSE:MDT) reported positive one-year, real-world clinical data for its MiniMed 780G insulin pump platform in children. Data came from a subset of 3,211 pediatric and adolescent patients (15 years old and below) with type 1 diabetes using the MiniMed 780G system with the Guardian Sensor 3 in Europe. According to a news release, the […]
Auto-injectors
Insulet announces board of director changes
Insulin delivery technology developer Insulet (NSDQ:PODD) announced today that it appointed Dr. Luciana Borio to its board of directors. Borio joins the board of Acton, Massachusetts–based Insulet, having served in key roles with the FDA, the National Security Council and the Council on Foreign Relations. She specializes in biodefense and medical countermeasures, emerging infectious diseases, […]
Insulet CEO Shacey Petrovic touts Omnipod 5, expects device launch this year
With FDA clearance imminent, Petrovic says Insulet’s latest device offers real change in diabetes care. Faced with either a bulky insulin pump or delivering multiple daily injections, Boston-based entrepreneur and venture capitalist John Brooks III sought an easier way to treat his young son’s newly diagnosed type 1 diabetes. He assembled a team of engineers […]
New BD syringe manufacturing lines are up and running in Nebraska
BD (NYSE:BDX) announced today that it increased its manufacturing capacity and domestic supply for syringes and needles. Franklin Lakes, N.J.-based BD’s new syringe and needle manufacturing lines, which were completed on an accelerated timeline, represent BD’s partnership with Department of Health and Human Services’ (HHS) Assistant Secretary for Preparedness and Response (ASPR). In July 2020, […]
FDA offers expanded clearance for DreaMed’s AI-based support system for type 2 diabetes
DreaMed Diabetes AI announced today that it received FDA clearance that expands its platform’s target population to cover type 1 and type 2 diabetes. Tel Aviv, Israel-based DreaMed’s Advisor Pro AI clinical decision support system assists healthcare providers in the management of diabetes patients who use insulin pumps or injections and monitor their glucose using […]
BREAKING: Medtronic expands Class I recall of MiniMed 600 series insulin pumps
The FDA today announced that Medtronic (NYSE:MDT) has expanded the Class I recall of its MiniMed 600 series insulin pumps. Medtronic today notified customers by email and phone that the recall has been expanded to replace all MiniMed 600 series insulin pumps that contain the clear container ring. The Fridley, Minnesota-based company first warned of […]
Welldoc wins expanded FDA clearance for BlueStar insulin dosing platform
Welldoc announced today that it received another FDA 510(k) clearance for its BlueStar diabetes treatment platform. Columbia, Maryland-based Welldoc’s new clearance for BlueStar — the ninth in total — expands insulin dosing support to most types of insulin, including bolus and premixed insulin titration for type 2 diabetes. Previous FDA clearances include in June 2020 […]
Alfred E. Mann Foundation sells insulin infusion pump IP to Medtronic
The Alfred E. Mann Foundation for Scientific Research (AMF) today announced the sale of intellectual property to Medtronic (NYSE:MDT). As part of the transaction of IP related to implanted infusion pumps, AMF will work with Medtronic to develop the technology into a next-generation implantable insulin pump for people living with hard-to-treat type 1 diabetes in […]
Diabeloop joins corporate advisory partnership, seeks to develop business in U.S.
Diabeloop announced today that it set up a local and dedicated team to support its pursuit of entry into the U.S. market. Paris-based Diabeloop develops an automated insulin delivery (AID) system that deployed throughout Europe this past year, making it commercially available in Germany, the Netherlands, Italy, Spain and Switzerland, with more countries to come. […]
Glaukos submits supplemental PMA application for iStent Infinite
Glaukos (NYSE:GKOS) announced today that it submitted a supplemental premarket approval application to the FDA for its iStent Infinite system. San Clemente, California–based Glaukos designed the iStent Infinite trabecular micro-bypass system for use in a standalone procedure to reduce elevated intraocular pressure (IOP) in patients with open-angle glaucoma uncontrolled by prior surgery or medical therapy. […]