Cook Medical’s Regentec division today announced the launch of a clinical trial to evaluate its HemaTrate blood filtration system to treat patients with critical limb ischemia (CLI) due to peripheral arterial disease (PAD). Indianapolis-based Cook Regentec said the HemaTrate system is designed to produce an autologous, peripheral blood-derived total nucleated cell (TNC) concentrate for intramuscular […]
Clinical Trials
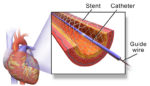
Cook Medical touts paclitaxel-coated stent PAD study results
Cook Medical today touted five-year data from a study into the use of its Zilver PTX paclitaxel-coated stent for peripheral arterial disease (PAD). The study concluded that there was no increase in long-term all-cause mortality due to paclitaxel after treatment using the Zilver PTX stent compared to traditional angioplasty or a bare-metal stent, according to a […]
Satsuma Pharmaceuticals registers $75m IPO
Satsuma Pharmaceuticals has registered an initial public offering, seeking to raise $75 million, according to a regulatory filing. The South San Francisco-based company is working on STS101, a nasal powder version of dihydroergotamine for migraine. It intends to use the money to fund its Phase 3 safety and efficacy clinical trials for STS101, manufacturing costs, […]
Medtronic’s In.Pact AV DCB meets endpoints in trial
Medtronic (NYSE:MDT) said last week that its clinical study comparing the In.Pact AV drug-coated balloon to percutaneous transluminal angioplasty (PTA) met primary safety and effectiveness endpoints. The study pitted the In.Pact AV DCB against PTA in patients with de novo or non-stented restenotic arteriovenous fistulae lesions. Results were presented at the Cardiovascular and Interventional Radiology Society […]
Concept Medical wins FDA breakthrough nod for MagicTouch AVF drug-coated balloon
Concept Medical said yesterday that it won breakthrough designation from the FDA for its MagicTouch AVF drug-coated balloon. MagicTouch AVF is designed to treat stenotic lesions of arteriovenous fistulae or arteriovenous graft in hemodialysis treatment of renal failure. India-based Concept Medical’s device is indicated for use in percutaneous transluminal angioplasty. The PTA procedure, using Concept […]
Surmodics reaps $10m from Abbott on SurVeil balloon milestone
Surmodics (NSDQ:SRDX) said today that it’s due for a $10 million milestone payment from Abbott (NYSE:ABT) after closing enrollment for the Transcend pivotal trial of its SurVeil drug-coated balloon for treating peripheral artery disease. Abbott in February 2018 paid $25 million up front for the global commercialization rights to the SurVeil device, which uses a proprietary […]
Study: No difference between Biotronik, Medtronic or Boston Scientific stents
A study comparing drug-eluting stents made by Biotronik, Medtronic (NYSE:MDT) and Boston Scientific (NYSE:BSX) found no statistically significant difference on target vessel failure or stent thrombosis after three years. The 3,514-patient Bio-Resort trial compared Biotronik’s Orsiro biodegradable sirolimus-eluting stent, Medtronic’s thin-strut zotarolimus-eluting Resolute Integrity stent and the Synergy biodegradable everolimus-eluting stent. Three-year results for some 3,393 […]
Medtronic wins FDA approval for new extended-wear infusion set
Medtronic (NYSE:MDT) said it earned investigational approval from the FDA to proceed with a trial for a new extended-wear infusion set. The goal of the study is to collect clinical data that supports the use of the device for up to seven days, more than twice the amount of time any infusion set can currently […]
Taris Bio, Bristol-Myers Squibb launch bladder cancer trial
Taris Biomedical has announced the dosing of the first patient in a study of its TAR-200 system plus a programmed immune checkpoint inhibitor for the treatment of muscle-invasive bladder cancer (MIBC). The TAR-200 system continuously delivers the chemotherapy drug gemcitabine in the bladder for multiple weeks. The combination with Bristol-Myers Squibb’s Opdivo (nivolumab) will be […]
Taris Biomedical touts results of bladder cancer treatment study
Taris Biomedical today announced positive results from the second arm of a study of its TAR-200 system for the treatment of patients with muscle-invasive bladder cancer (MIBC). The most recent study arm involved 10 patients treated with TAR-200 and yielded positive safety, tolerability and preliminary efficacy data, according to Lexington, Mass.-based Taris. Combined data from […]