Glenmark Pharmaceuticals said today that the FDA approved its Investigational New Drug application for a Phase II study of its nebulized tiotropium bromide. The Mahwah, N.J.-based company plans to enroll 155 patients with mild to moderate chronic obstructive pulmonary disease and evaluate its GSP 304 formulation as a daily maintenance treatment for bronchospasm. “Respiratory is a core […]
Clinical Trials
PharmaJet, Serum Institute partner for needle-free MMR vaccine
PharmaJet inked a partnership with vaccine manufacturer Serum Institute of India to commercialize needle-free delivery of the MMR vaccine using the Stratis 0.5mL injection system. PharmaJet’s device is designed to administer medication intramuscularly or subcutaneously, without the use of needles. The Chicago-based company touts the technology as a way to prevent cross-contamination between needles and enhance patient compliance. […]
Mast Therapeutics subsidiary advances trial for inhaled sodium nitrite solution
Mast Therapeutics (NYSE:MSTX) said today that its subsidiary Aires Pharmaceuticals inked a deal with the University of Pittsburgh related to its phase 1/2 open-label proof-of-concept trial for the company’s AIR001 inhaled sodium nitrite solution in patients with cystic fibrosis. The study will evaluate the nebulized drug as a treatment of Pseudomonas aeruginosa infection in CF […]
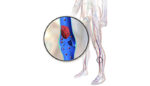
Study evaluates drug-device combo therapy for deep vein thrombosis
A National Institutes of Health-sponsored study found that most patients with deep vein thrombosis should be treated with anticoagulant drugs alone, without a procedure-based intervention. However, the same study showed that a minimally-invasive catheter-director therapy, pharmacomechanical catheter-directed thrombolysis, provided greater initial pain relief and could prevent disability in DVT patients. The research was presented today […]
InGeneron raises $20m in Series D for regenerative stem cell therapies
InGeneron said today that healthcare provider Sanford Health invested $20 million in the company’s Series D financing round. The Houston, Texas.-based company said the funding will be used to support the clinical programs for its stem cell injections in patients with rotator cuff and venous ulcer tears. “This significant investment demonstrates Sanford’s commitment to be […]
Medtech stories we missed this week: March 3, 2017
Several clinical study results were published this week, while other companies reached distribution deals. Here are some medtech stories we missed this week but were still worth mentioning. 1. Health Canada approves Ventripoint’s complete heart analysis system for 2D ultrasounds Ventripoint Diagnostics received a license from Health Canada for its new VMS-Plus machine and the 4-chamber (4C) heart […]
Researchers develop micropump for patients with glaucoma
Researchers from the University of Southern California’s Roski Eye Institute developed a micropump that can be implanted directly into the eye and deliver a predetermined dose of the diabetic macular edema drug ranibizumab. Current treatments for glaucoma are traditionally delivered using eye drops. But studies show that some patients stop taking their prescription glaucoma eye […]
Aerie resubmits NDA for Rhopressa eye drops
Aerie Pharmaceuticals (NSDQ:AERI) said today that it resubmit a New Drug Application for the company’s Rhopressa eye drops. The Irvine, Calif.-based company’s NDA was withdrawn in October last year, after a contract manufacturer was not prepared for its pre-approval inspection. Aerie’s Rhopressa eye drops specifically target the eye’s trabecular network – the diseased tissue responsible for […]
BioCardia, Johns Hopkins treats first patient with CardiAmp cell therapy in Phase III trial
Johns Hopkins, the Maryland Stem Cell Research Fund and BioCardia (NSDQ:BCDA) said today that the 1st patient has been treated in a pivotal Phase III trial of the CardiAmp cell-based therapy for the treatment of ischemic heart failure. The investigational therapy was designed to deliver a dose of a patient’s own bone marrow cells directly […]
Gamida Cell touts first patient treated in Phase III NiCord trial
Gamida Cell said today that the 1st patient has been treated in the Phase III registration trial of its NiCord cell graft for patients with blood cancer. The treatment is a graft derived from umbilical cord blood which has been expanded and enriched with stem and progenitor cells, according to the company. The Jerusalem-based company’s […]