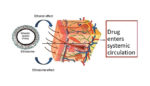
Ethosomes – nanocarriers loaded with ethanol – can penetrate the skin’s protective layers and deliver a variety of drugs, a review published in Current Clinical Pharmacology reported today. Transdermal delivery of drugs is touted by some researchers as promising, since it’s noninvasive and avoids any interaction with the gastrointestinal tract. Ethosomes are malleable and their […]
Clinical Trials
Dicerna bails on EnCore lipid nanoparticle tumor delivery program
Dicerna Pharmaceuticals (NSDQ:DRNA) said yesterday that it plans to spike the development program for its lipid nanoparticle delivery system for solid tumors because its preliminary results don’t meet expectations. The Cambridge, Mass.-based company evaluated its lipid nanoparticle, DCR-MYC, in a pair of clinical trials – a Phase I study of patients with solid tumors, hematological […]
Changing healthcare & clinical trials with patient monitoring tech
Patient monitoring can be challenging, often relying on patients to self-report symptoms or describe how they’re feeling. But technology is changing that, making it easier for researchers to monitor patient adherence to a trial’s regimen and for doctors to personalize a patient’s treatment. At the FierceBiotech Drug Development Forum in Boston today, a panel took up the […]
FDA approves clinical trial for QT Vascular’s Chocolate Touch DCB
QT Vascular said today that it won an investigational device exemption from the FDA for a pivotal study of its Chocolate Touch drug-coated balloon. The device won the CE Mark for treating peripheral artery disease in September 2015 and in July it won approval for treating coronary artery disease. The Singapore-based company is slated to […]
FDA approves 6-month endpoint in trial of Bard’s Lutonix 014 drug-coated balloon
C.R. Bard (NYSE:BCR) said yesterday that the FDA approved a supplemental investigational device exemption to change the main 6-month endpoint in the clinical trial of a new version of its Lutonix drug-coated balloon in treating below-the-knee peripheral arterial disease. Murray Hill, N.J.-based Bard in October 2014 won pre-market approval from the FDA for the Lutonix 035 DCB for PAD lesions above the […]
OptiNose touts Phase III data for exhaled breath drug delivery
OptiNose touted data from a pair of Phase III clinical trials proving the safety and efficacy of its exhaled breath drug delivery treatment for chronic rhinosinusitis patients with and without polyps. The company presented the data today at the American Rhinologic Society’s annual meeting in San Diego. Data from both trials showed limited adverse effects from […]
Ablative Solutions wins FDA nod for Phase II chemical denervation trial
Ablative Solutions said today that the FDA approved a Phase II trial of its chemical denervation system for hypertension under its investigational new drug application. The trial, called Target-BP I, will evaluate the efficacy of Ablative’s Peregrine infusion ablation system, which uses ethanol to interrupt signals from sympathetic nerves in the renal artery to control […]
Novocure touts long-term analysis of Optune drug-device combo
Novocure (NSDQ: NVCR) touted a long-term analysis of data from a Phase III pivotal trial of its Optune device in combination with temozolomide for the treatment of newly-formed glioblastomas. Survival data for nearly 700 patients confirmed interim results from the trial, 1st published in December 2015 in the Journal of the American Medical Assn. Patients treated […]
More Respimat data from Boehringer
Boehringer Ingelheim on Tuesday touted results from the Phase III CanoTinA asthma trial, which showed that adding tiotropium Respimat to maintenance asthma therapy in kids ages 6-11 significantly improves lung function compared to placebo. The results were presented Tuesday at the European Respiratory Society International Congress 2016 in London. This comes 1 day after results from another clinical trial […]
JDRF backs Sernova’s implant for T1D
Canadian-based drug device maker Sernova Inc. is working with the Juvenile Diabetes Research Foundation on a clinical trial of Sernova’s cell pouch system to treat hypoglycemia unawareness patients with severe type 1 diabetes. The JDRF will give $2.45 million to support a clinical trial at a transplantation center in the U.S., according to the agreement. Hypoglycemia […]