Monaghan Medical yesterday touted data from a 6-month retrospective study showing that its Aerobika device reduced drug use for treatment of chronic obstructive pulmonary device. The Syracuse, N.Y.-based company says its oscillating positive expiratory pressure therapy device is designed to expand the airways, help expel mucus to the upper airways and aid in drug deposition. The […]
Drug-Device Combinations
Kaleo plans 2017 U.S. relaunch for EpiPen competitor Auvi-Q
Kaléo Inc. said today that it plans to relaunch its Auvi-Q injector for emergency allergy treatment in the U.S. during the 1st half of 2017, according to Reuters. The Richmond, Va.-based company’s device was recalled last year after concerns about the accuracy of delivered dose. Sanofi (NYSE:SNY) made and licensed the Auvi-Q device from Kaléo, but returned rights to […]
PharmaJet, Vaccibody collaborate on needle-free HPV vaccine
PharmaJet and Vaccibody inked a collaborative deal to provide needle-free injection tech for the development of a human papillomavirus vaccine. HPV is the cause of virtually all cervical cancer, according to the National Cancer Institute, and just 2 types of HPV, 16 and 18, are responsible for about 70% of all cases. PharmaJet’s device is […]
Medtronic secures Health Canada license for MiniMed 630G system
Medtronic (NYSE:MDT) subsidiary Medtronic Canada said today that it landed a license with Health Canada for its MiniMed 630G insulin pump system for patients with diabetes mellitus. The Fridley, Minn.-based company said it interviewed more than 1,000 diabetes patients to better understand the features they look for in an insulin pump. The final product has a […]
Sunovion, medical society ally for COPD drug-delivery survey
Sunovion Pharmaceuticals and the American College of Chest Physicians yesterday launched a strategic initiative called Delivery Makes a Difference to better understand patient and healthcare provider practices in COPD therapy. A series of “nationally recognized COPD thought leaders” came together to be a part of the steering committee for the initiative. They developed a survey for 1,000 […]
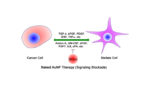
Gold nanoparticles hold promise for pancreatic cancer treatment
Gold nanoparticles could be an important part of developing a treatment for pancreatic cancer, according to new research published yesterday in the journal ACS Nano. Previous work demonstrated that gold nanoparticles can be an effective vehicle to carry chemotherapy drugs into tumors. They can also act as a target to enhance the impact of radiation on […]
Mylan, Theravance touts Phase III data for COPD drug
Mylan (NSDQ:MYL) and Theravance Biopharma, Inc. (NSDQ:TBPH) said today that revefenacin, the 1st nebulized bronchodilator in development for the treatment of chronic obstructive pulmonary disease, demonstrated positive results in 2 replicate phase III efficacy studies. Data from more than 1,250 moderate to severe COPD patients showed that both studies met their efficacy and safety endpoints. Revefenacin […]
Akesys, Elixir Medical tout 1st implant for Prava bioresorbable peripheral stent
Akesys Medical and Elixir Medical Corporation said today that it implanted its 1st Prava sirolimus eluting bioresorbable peripheral scaffold for the treatment of blockages in the superficial femoral artery. The scaffold was developed to keep the large artery in the thigh open and restore blood flow to the leg and resorb into the body, making […]
SteadyMed details commercialization plans for Trevyent
SteadyMed Therapeutics (NSDQ:STDY) today detailed the development of its lead drug candidate, Trevyent (treprostinil sodium), for the treatment of pulmonary arterial hypertension using its PatchPump tech at the Parenteral Drug Assn. Conference in Huntington Beach, Calif. PatchPump is the 1st single-use, wearable, pre-filled pump and treprostinil will be the 1st drug integrated with the delivery system, according to the San […]
FDA awards product development grants for rare diseases
The FDA said yesterday that it awarded 21 clinical trial research grants totaling more than $23 million over the next 4 years to stimulate product development for patients with rare diseases. The Orphan Products Clinical Trials Grants Program has awarded more than $370 million to fund more than 590 clinical studies since its creation in […]