Glaukos (NYSE:GKOS) announced today that it submitted a New Drug Application (NDA) to the FDA for its iDose TR implant. Aliso Viejo, California-based Glaukos designed iDose TR as a micro-invasive intraocular implant. It continuously delivers therapeutic levels of a proprietary formulation of travoprost from within the eye over extended periods of time. Once all travoprost […]
Food & Drug Administration (FDA)
FDA committee unanimously votes in favor of over-the-counter Narcan from Emergent BioSolutions
Emergent BioSolutions (NYSE:EBS) announced today that its over-the-counter Narcan (naloxone HCl) nasal spray received unanimous approval from governing bodies. The vote came from the FDA Nonprescription Drugs Advisory Committee and the Anesthetic and Analgesic Drug Products Advisory Committee. A total of 19 voters decided in favor of the benefit-risk profile for Narcan. The voters support […]
Tidepool Loop automated insulin delivery system wins FDA clearance
Tidepool announced recently that the FDA cleared its Tidepool Loop, a “do-it-yourself,” (DIY) automated insulin delivery (AID) system. Palo Alto, California-based Tidepool is a non-profit diabetes data management group. It created the hybrid, closed-loop AID app for compatibility with multiple insulin pumps and continuous glucose monitors. The company announced collaborations in the past that include […]
Know Labs inks partnerships to accelerate progress toward FDA clearance for glucose monitor
Know Labs (NYSE:KNW) announced that it entered into a series of strategic partnerships as it looks to move toward a major regulatory nod. Seattle-based Know Labs develops the Bio-RFID technology. It uses spectroscopy to direct electromagnetic energy through a substance or material to capture a unique molecular signature. This technology integrates into wearable, mobile or bench-top […]
Vero Biotech wins FDA approval for inhaled nitric oxide delivery with anesthesia in the OR
Vero Biotech announced that the FDA approved its second-generation Genosyl system for use with rebreathing anesthesia in the operating room setting. Atlanta-based Vero Biotech designed Genosyl for inhaled nitric oxide (iNO) delivery. This approval covers the second-generation Genosyl device. The third-generation device — which recently received FDA approval — has not yet been tested with […]
The year ahead in diabetes care: what to expect in 2023
After a banner year for diabetes technology in 2022, there’s still plenty to look forward to in 2023. Throughout 2022, we saw a wide range of advancements in diabetes technology. We saw launches for next-generation technologies, exciting clinical trial results, rumored spinouts and acquisitions and more. You can read all about the biggest diabetes stories […]
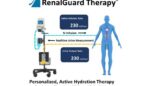
CardioRenal Systems wins FDA breakthrough nod for RenalGuard Therapy device
CardioRenal Systems announced today that it received FDA breakthrough device designation for its RenalGuard Therapy device. Milford, Massachusetts-based CardioRenal Systems designed the device for preventing acute kidney injury (AKI). It’s for patients at risk for cardiac surgery-associated AKI. RenalGuard protects the kidneys with personalized, active hydration. The system maximizes urine output while balancing hydration. It […]
Surmodics announces regulatory delay for its SurVeil balloon
The FDA noted more information on biocompatibility and labeling is needed.
The FDA has indicated that Surmodics’ Surveil drug-coated balloon premarket approval application is not approvable. According to an FDA letter to the company, Surmodics’ Surveil was not approved due to certain information in the biocompatibility and labeling categories. The governing agency said more information needs to be added by an amendment to Surmodics’ PMA application to […]
How Dexcom aims to extend its mission beyond diabetes
CEO Kevin Sayer explains how, following the launch of the next-generation Dexcom G7, the company is looking toward the future. Dexcom (Nasdaq:DXCM) ended 2022 strong, receiving FDA clearance for its next-generation G7 continuous glucose monitor (CGM). With a U.S. launch imminent, the company could keep its efforts centered on the diabetes space. Instead, the company […]
MedAlliance picks up FDA IDE nod for drug-eluting balloon
MedAlliance announced today that its novel sirolimus-eluting balloon received conditional FDA investigational device exemption (IDE). Geneva, Switzerland-based MedAlliance can now initiate its pivotal clinical trial for the treatment of coronary de novo lesions. MedAlliance received its first IDE for Selution SLR in below-the-knee indications in May 2022. It picked up a second IDE in August. […]