Gamida Cell said today that the European Medicines Agency granted its NiCord cell graft an additional orphan drug designation as a treatment for hematopoietic stem cell transplantation, or bone marrow transplantation. The treatment is a graft derived from umbilical cord blood which has been expanded and enriched with stem and progenitor cells, according to the […]
Oncology
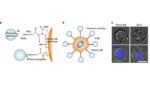
Study: Tethered nanoparticles help trigger cell death in cancerous tumor cells
Researchers from the Massachusetts Institute of Technology have developed a way to amplify certain types of cancer treatment by tethering nanoparticles to cancerous tumor cells. The team’s work was published in Nature Communications. The researchers found that tethered nanoparticles increase the forces exerted on the cells by phenomena such as blood flow, therefore making the cells […]
Penn State researchers deliver melanoma drug combo using nanoparticles
Researchers from the Penn State College of Medicine developed a method to deliver a combination of drugs using nanoparticles in the hopes of preventing melanoma cancer cells from developing treatment-resistance. Their work was published in Molecular Cancer Therapeutics and Cancer Letters. The team’s CelePlum-777 localized around cancer tumors and delivered a specific ratio of an anti-inflammatory, Celecoxib, and a toxin, Plumbagin, […]
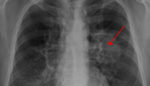
New blood-based test could detect all lung cancer types
Exact Sciences and the Mayo Clinic recently collaborated on a blood-based lung cancer test that researchers say has shown high accuracy for detecting all stages of lung cancer. “These results reveal an opportunity to detect lung cancer from a simple blood draw,” said Kevin Conroy, chairman and CEO of Exact Sciences, in a news release. “Our […]
BTG wins CE Mark for DC Bead Lumi radiopaque drug-eluting bead
BTG (LON:BTG) said today that it won CE Mark clearance in the European Union for its DC Bead Lumi radiopaque drug-eluting bead. The bead is designed to be loaded with doxorubicin or irinotecan to locally treat tumors in patients with hepatocellular carcinoma and malignant colorectal cancer metastasised to the liver. The company’s DC Bead Lumi also […]
EnGeneIC wins orphan drug designation for targeted nanocells
EnGeneIC said today that the FDA granted orphan drug designation to the company’s targeted EDV nanocells for the treatment of glioblastoma multiforme. The clinical stage biopharmaceutical company is developing bacterially-derived EDV nanocells as a nanoparticle, siRNA and miRNA delivery platform designed to target and kill tumor cells, while triggering the immune system’s natural anti-tumor response. […]
Gamida Cell touts first patient treated in Phase III NiCord trial
Gamida Cell said today that the 1st patient has been treated in the Phase III registration trial of its NiCord cell graft for patients with blood cancer. The treatment is a graft derived from umbilical cord blood which has been expanded and enriched with stem and progenitor cells, according to the company. The Jerusalem-based company’s […]
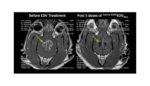
OncoSec wins fast track designation for electroporation combo therapy
OncoSec Medical (NSDQ:ONCS) said today that it won Fast Track Designation from the FDA for its ImmunoPulse IL-12 electroporation gene therapy for the treatment of metastatic melanoma. The biotech company’s electroporation device is designed to locally deliver DNA-based interleukin-12 to stimulate the immune system and fight off cancer cells. The ImmunoPulse device delivers a sequence […]
Novocure enrolls 1st patient in TTFields, chemo treatment lung cancer phase III trial
Novocure (NSDQ:NVCR) said today it enrolled the 1st patient in the phase 3 pivotal Lunar trial of its Tumor Treating Field technology for treating patients with advanced non-small cell lung cancer who have failed platinum-based therapy. The prospective, randomized 512-patient trial from the St. Helier, N.J.-based company aims to test the safety and efficacy of its […]
Immunomedics, Seattle Genetics ink $2B deal for antibody-drug conjugate
Immunomedics (NSDQ:IMMU) said it landed an exclusive global licensing agreement with Seattle Genetics (NSDQ:SGEN) for Immunomedics’ antibody-drug conjugate, IMMU-132, for the treatment of solid tumors. The deal could potentially bring in $2 billion plus royalties for Immunomedics. According to the deal, Seattle Genetics is responsible for developing, funding, manufacturing and commercializing IMMU-132. The Washington-based company will […]