(Image from Lyra Therapeutics)
Lyra Therapeutics (NSDQ:LYRA) presented positive results from a Phase 2 study of its therapeutic for treating chronic rhinosinusitis (CRS).
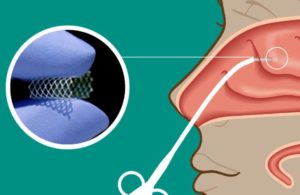
Watertown, Mass.-based Lyra’s Phase 2 Lantern study evaluated its LYR-210 long-acting product for CRS, which is administered in-office to deliver a sustained release for up to six months as a non-invasive alternative to surgery for patients who have failed medical management.
LYR-210 is delivered through the XTreo platform, comprised of a flexible mesh capable of taking a high payload of a drug and delivering to difficult-to-access nasal inflammation sites.
Read an interview with executives about Lyra Therapeutics’ approach to treating CRS here.
Topline data presented at the Combined Otolaryngology Spring Meetings (COSM) 2021 included 24-week data that highlighted improvement from baseline in bilateral ethmoid Zinreich scores by MRI, symptom improvement in both polyp and non-polyp patients and the need for and use of rescue medication during the trial.
“The data presented today at COSM demonstrates LYR-210’s rapid and durable dose-dependent improvement based on several outcome measures, including the cardinal symptoms of CRS, SNOT-22, and MRI, in both non-polyp and polyp patients, from a single in-office administration,” principal investigator & Garnett Passe & Rodney Williams Memorial Foundation Chair in Otolaryngology at the University of Queensland Anders Cervin said in the release. “Based on the results of the Lantern trial, and my own experience with the drug candidate, I believe that LYR-210 has the potential to represent a major step forward in the care for CRS patients who are facing surgery as their next treatment option.”
Patients in the trial, both in polyp and non-polyp populations, achieved the minimally clinically important difference in the trial, while just one patient in the 7500µg-dose group and two patients in the 2500µg-dose group required a rescue treatment compared to seven patients in the control over 24 weeks.
Lyra also shared an analysis of Lantern that focused on a composite of the three cardinal symptoms of CRS, confirming that LYR-210 achieved statistically significant improvement compared to control in treating nasal blockage, nasal discharge and facial pain.
“We are delighted to present the full data set from our Lantern study at COSM. I believe today’s data supports LYR-210’s ability to provide up to 24 weeks of effective symptom relief,” Lyra CMO Dr. Robert Kern said. “LYR-210 is designed to provide continuous and consistent steroid treatment directly to diseased tissue and, additionally, eliminate patient compliance challenges. The additional data presented today shows that LYR-210 decreased ethmoid opacification and the need for rescue treatment in the Lantern study, providing yet further evidence for LYR-210’s efficacy beyond the previously announced 4CS and SNOT-22 endpoints.”
“Our successful Lantern study was designed to inform a pivotal Phase 3 trial for LYR-210, and today’s full data set has further shaped our thoughts regarding a preferred design for that trial, subject of course to an End-of-Phase 2 meeting with the FDA,” added Lyra president & CEO Maria Palasis. “In order to best address a combined polyp and non-polyp population, we are currently considering efficacy endpoints based on a composite 3CS of CRS score for our pivotal study, with the aim of bringing a much-needed new therapy to CRS patients as expeditiously as possible.”

