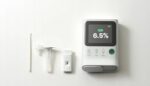
Fractyl Health announced that it initiated its Revita Global Registry Program in Germany to study its diabetes reversal treatment. On April 20, the Ethics Committee of the University of Freiburg (Germany) granted clearance for the initiation of a registry study. The study covers the Revita system in German hospitals. Patients in the registry have inadequately […]
Diabetes
FDA clears basal-only insulin pod for type 2 diabetes from Insulet
Insulet (Nasdaq:PODD) announced today that it received FDA clearance for its Omnipod GO long-acting insulin delivery device. Omnipod GO, which received clearance for people with type 2 diabetes aged 18 or older, covers the basal-only insulin population. The target population typically takes daily injections of long-acting insulin. The first-of-its-kind, standalone, wearable insulin delivery system provides […]
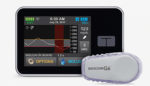
FDA approves next-gen MiniMed 780G insulin pump from Medtronic
Medtronic (NYSE:MDT) announced that it received FDA approval for its MiniMed 780G system with the Guardian 4 sensor. The news — posted in the evening on April 21 — has MDT shares up more than 4% to $89.36 apiece in afternoon trading today. (MassDevice’s MedTech 100 Index is up slightly.) The positive news for the […]
Study supports non-invasive glucose monitoring tech from Know Labs
Know Labs (NYSE:KNW) today announced positive study data supporting its proprietary non-invasive glucose monitoring technology. Seattle-based Know Labs develops its proprietary Bio-RFID technology. It uses spectroscopy to direct electromagnetic energy through a substance or material to capture a unique molecular signature. This technology integrates into wearable, mobile or bench-top form factors. Know Labs developed the first […]
Orange Biomed unveils at-home A1c test in the U.S.
Orange Biomed announced today that it commenced a U.S. launch as it looks to fill testing gaps for pre-diabetes and diabetes. Seoul, South Korea–based Orange Biomed operates out of Research Triangle Park, North Carolina in the U.S. The company currently has ongoing studies for its OBM rapid A1c test. It designed the glycated hemoglobin analyzer […]
Senseonics announces first pediatric 365-day CGM insertion
Senseonics (NYSE:SENS) announced that the first pediatric study participant received an Eversense 365-day CGM insertion. The insertion took place as part of the company’s ENHANCE clinical trial at the AMCR Institute. AMCR, a clinical research center, focuses on pre-diabetes, type 1, type 2 diabetes and obesity. The study operates under the direction of Dr. Timothy […]
Dexcom, Abbott applaud Medicare expansion for CGMs
Dexcom (Nasdaq:DXCM) and Abbott (NYSE:ABT) both commended a change to Medicare policies that benefits continuous glucose monitor (CGM) users. Last month, the Centers for Medicare & Medicaid Services (CMS) published a final local coverage determination (LCD) expanding coverage for CGMs. The ruling has now gone into effect. CMS first published the LCD modifying coverage criteria for […]
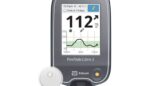
Tandem reports positive research for its artificial pancreas tech
Tandem Diabetes Care (Nasdaq:TNDM) today announced a positive meta-analysis of three randomized controlled trials of its Control-IQ advanced hybrid closed-loop technology. The research found immediate and sustained improvements for a diverse range of subjects using Control-IQ technology, according to the research published in the journal Diabetes Technology & Therapeutics. There was an average increase in […]
Abbott breaks ground on FreeStyle Libre manufacturing plant in Ireland
Abbott (NYSE:ABT) today broke ground on a new FreeStyle Libre manufacturing facility in Kilkenny, Ireland. Jared Watkin, SVP for Abbott’s diabetes care business, shared the news of the groundbreaking in a LinkedIn post. In August 2022, Ireland announced Abbott’s plans to spend €440 million ($450 million) to expand its operations in Ireland. The investment included […]
FDA clears reader for next-gen Abbott FreeStyle Libre 3
Abbott (NYSE:ABT) announced today that the FDA cleared a reader device for its FreeStyle Libre 3 continuous glucose monitoring (CGM) system. The FDA cleared Abbott’s next-generation FreeStyle Libre 3 in May 2022. Abbott designed FreeStyle Libre 3 as the smallest and thinnest CGM sensor in the world. The system constitutes the size of two stacked […]