Democrats from the House Committee on Oversight and Government Reform have sent letters to seven drugmakers as part of an investigation into why prices for multiple sclerosis drugs have nearly quadrupled since 2004. Reps. Elijah Cummings (D-Md.) and Peter Welch (D.-Mass) penned inquiries to Bayer (ETR:BAYN), Biogen (NSDQ:BIIB), EMD Serono, Novartis (NYSE:NVS), Roche (OTC:RHHBY), Sanofi Aventis (NYSE:SNY) and Teva Pharmaceuticals (NYSE:TEVA). […]
Featured
Mylan inks $465m settlement with DOJ over EpiPen Medicaid classification
Mylan (NSDQ:MYL) said today that it inked a $465 million deal with U.S. Dept. of Justice to settle claims that it misclassified its EpiPen allergy auto-injector device with the Medicaid Drug Rebate Program. In October last year, The Centers for Medicare & Medicaid Services said that Mylan had been overcharging Medicaid for its EpiPen device for years, […]
Alzeca Biosciences raises $11m for Alzheimer’s diagnostic imaging agent
Alzeca Biosciences said today that it raised $11 million in a Series A round to fund the development of its MRI imaging agent designed for the early diagnosis of Alzheimer’s disease and other neurodegenerative disorders. The company touts its ADx nanoparticle as the first MRI contrast agent to target amyloid – a brain protein linked […]
Novo Nordisk diabetes drug succeeds in late-stage trial
Shares in Novo Nordisk (NYSE:NVO) rose nearly 5% yesterday afternoon after the insulin-maker said that its once-weekly diabetes drug, semaglutide, succeeded in lowering glucose levels in a late-stage trial. The 40-week trial enrolled 1,200 patients and evaluated two dosages of semaglutide in combination with a standard-of-care-drug. The study compared its drug against Eli Lilly‘s (NYSE:LLY) dulaglutide […]
7 diagnostic devices to boost healthcare in the developing world
The World Health Organization estimates that a quarter of death and disease globally is caused by hazards and environmental burdens in developing countries with little to no access to preventative care and diagnostic devices. Since developing countries are poor agricultural regions that are still becoming economically and socially advanced, it is harder for doctors to […]
Update: Industry panels disintegrate in wake of Trump remarks
A pair of industry panels convened by President Donald Trump disintegrated today in the wake of his latest comments on the racist violence in Charlottesville. After Merck (NYSE:MRK) CEO Kenneth Frazier announced on Monday that he planned to leave Trump’s manufacturing advisory panel, a wave of executives followed in his path. Today, reversing from a statement […]
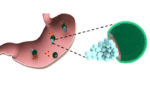
Study: Drug-delivery micromotors treat bacterial infection in stomach
Researchers at the University of California San Deigo have successfully used tiny micromotors loaded with antibiotics to treat a bacterial infection in the stomachs of mice, according to a study published today in Nature Communications. The tiny drug-delivery vehicles are powered by gastric acid, pushing them through the stomach and releasing cargo in a specific region, […]
Roche, Mimetas develop “gut-on-a-chip” tech for drug-testing
Mimetas said today that it collaborated with Roche (OTC:RHHBY) to develop an organ-on-a-chip system that is designed to evaluate intestinal permeability in 3D gut tubules following exposure to drug compounds. The results of the collaboration were published in Nature Communications. The researchers reported that the intestinal tubules were leak-tight after just four days in culture and […]
Glytec lands another 510(k) clearance for diabetes management software
Glytec said today that it won a fourth 510(k) clearance from the FDA for its insulin titration software, eGlycemic Management System featuring Glucommander. The latest FDA nod applies to product enhancements derived from user feedback, the Waltham, Mass.-based company said, including a streamlined transition of inpatients from IV to subcutaneous therapy and more robust workflow […]
Amneal Pharmaceuticals issues voluntary recall of lorazepam droppers
Amneal Pharmaceuticals said today that it is voluntarily recalling 13 lots of its oral lorazepam product due to a defect in the dosing dropper. In some cases, the dropper is printed with the dose markings in reverse order or has no dose markings at all, according to the Bridgewater, N.J.-based company. So far, no adverse […]