(Image from Lyra Therapeutics)
Lyra Therapeutics (Nasdaq:LYRA) announced today that it enrolled the first patient in its Phase 3 Enlighten II clinical trial.
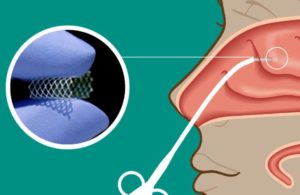
Enlighten II is evaluating the company’s LYR-210 in adult patients with chronic rhinosinusitis (CRS). Lyra’s proprietary XTreo platform delivers LYR-210.
Watertown, Massachusetts-based Lyra designed the proprietary XTreo platform for precise, sustained and local delivery. The platform is comprised of a flexible mesh capable of taking a high payload of a drug.
XTreo provides a consistent release of the therapeutic over time, with Lyra touting capabilities of six months of drug delivery.
Enlighten II is the second Phase 3 trial in the pivotal clinical program for LYR-210. Lyra initiated Enlighten 1 in January. Both trials will enroll surgically naïve CRS patients in the U.S. and Europe to support the company’s FDA submission.
“We are excited about the ENLIGHTEN pivotal program, with both Phase 3 trials now actively enrolling patients,” said Maria Palasis, president and CEO of Lyra Therapeutics. “LYR-210 is the first CRS product candidate designed to provide six months of continuous therapy with a single treatment, and we are focused on the opportunity to bring a new standard of care to the millions of CRS patients suffering with the disease.”
Lyra’s Enlighten program
The Enlighten program includes a total of 360 adult CRS patients. Enlighten II will enroll approximately 180 CRS patients who have failed medical management and have not had prior sinus surgery. Patients will be randomized, 2:1, to either LYR-210 or control. The primary endpoint is the change from baseline in a composite score of three cardinal symptoms (3CS) of CRS (i.e., nasal blockage, nasal discharge, and facial pain) at week 24.
Tandem Clinical Research in Marrero, Louisiana, enrolled the first patient.
“I am very happy that the first patient has now been enrolled at our site in this important trial,” said Dr. Adil Fatakia, Tandem Clinical Research. “My investigator colleagues at other sites in the U.S. and around the world look forward to enrolling this trial to allow the required data to be collected, which will support submission of this program for approval.”

