 A team of researchers from the Massachusetts Institute of Technology and Brigham & Women’s Hospital have developed a drug-delivery capsule that can deliver a week’s worth of HIV drugs in a single dose.
A team of researchers from the Massachusetts Institute of Technology and Brigham & Women’s Hospital have developed a drug-delivery capsule that can deliver a week’s worth of HIV drugs in a single dose.
The technology is designed to make it easier for people with HIV to deal with the strict schedule of medications they need to take to fight the virus, according to a study published in Nature Communications this month.
“One of the main barriers to treating and preventing HIV is adherence,” Giovanni Traverso, an MIT researcher & gastroenterologist and biomedical engineer at Brigham & Women’s Hospital, told MIT News. “The ability to make doses less frequent stands to improve adherence and make a significant impact at the patient level.”
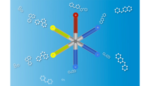
The team’s capsule contains a structure with six drug-loaded arms. After a patient swallows the capsule, the structure’s arms unfold and slowly release the drugs. The team has previously shown that the capsules can stay in the stomach for up to two weeks.
The product’s first version was made from a single polymer that carried whatever drug was to be delivered to the patient’s system. The polymer was also designed to provide structural support. This presented a problem to researchers – if they wanted to design structures that could deliver drugs at different rates, any changes to the polymer could impact the capsule’s ability to remain intact.
In the latest version, researchers created a capsule that has a strong polymer for its backbone, with six arms made of different drug-loaded polymers.
“In a way, it’s like putting a pillbox in a capsule. Now you have chambers for every day of the week on a single capsule,” Traverso explained.
The researchers evaluated the capsules in pigs and found that the device successfully made its way to the stomach and delivered three HIV drugs over the course of one week. After all of the drug has been delivered, the capsule disintegrates and pass through the animal’s digestive tract.
The company developing this drug-delivery technology, Lyndra, is reportedly working on taking the product to clinical trials.


I am Henry from Uganda,I havebeen impressed by the work you are doing to improve thelife of people living with HIV. I am a person living with HIV with my wife also posivive withHIV.I would like to access the onec a week pill to make our life better, how can I get it and how much will it cost us. I thank you very much for your positive response