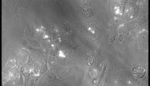
University of California San Diego researchers have created a technique that speeds up and slows down human heart cells that are being grown in a dish on command by shining light on them and varying the intensity. The heart cells are being grown in graphene which turns light into electricity, a more realistic environment that […]
Medtech stories we missed this week: June 8, 2018
From Royal Philips receiving FDA clearance to NeuroPace launching its epilepsy treatment device, here are seven medtech stories we missed this week but thought were still worth mentioning. 1. FDA clears Philips’s Ingenia Elition 3.0T MR scanner Royal Philips announced in a June 5 press releasethat it has received FDA 510(k) clearance for its Ingenia Elition […]
FDA plans fast-track development and marketing program for medical devices that treat opioid addiction
By Stewart Eisenhart, Emergo Group Medical device regulators at the US Food and Drug Administration have initiated a new campaign to support development and commercialization of novel devices aimed at pain treatment and opioid addiction. Get the full story here at the Emergo Group’s blog. The opinions expressed in this blog post are the author’s […]
FDA takes cautious approach to new regulations for OEM and third-party device servicers
By Joel Domanowski, Emergo Group The US Food and Drug Administration has cited insufficient evidence regarding public health concerns to warrant new regulatory requirements for original equipment manufacturers (OEM) and third-party servicers of medical devices, but will take less formal steps to ensure safety and effectiveness of these entities’ products and services. Get the full […]
Medtech stories we missed this week: May 25, 2018
From Mauna Kea receiving FDA clearance to Medacta getting clearance for its pedicle screw placement guide, here are seven medtech stories we missed this week but thought were still worth mentioning. 1. Mauna Kea wins FDA nod for neurosurgery indication Mauna Kea announced in a May 24 press release that it has received FDA 510(k) clearance […]
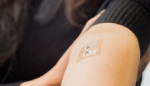
This tattoo-like sensor measures blood glucose levels non-invasively
Diabetes tracking can be a scary and tedious task, but University of California at San Diego researchers have developed a needless glucose monitor tattoo sensor that measures insulin levels through sweat on the skin. There are approximately 30.3 million people living with diabetes in the U.S., according to the American Diabetes Association. Monitoring blood sugar […]
FDA is asking for more information on application forms: Here’s why that’s good for innovation and improving health
By: Dr. Christopher Leptak To be most effective, electronic health records (EHRs) use a systematized and standardized nomenclature for the hundreds of thousands of clinical terms that characterize patient care. This helps to ensure consistency from EHR to EHR and allows these records to be usable from healthcare entity to health care entity, a concept […]
Brazil Anvisa changes market pathway for low-risk medical devices
By Stewart Eisenhart, Emergo Group Brazilian medical device regulators have approved plans to amend registration requirements for low-risk devices to more closely align with International Medical Device Regulators Forum (IMDRF) recommendations. Get the full story here at the Emergo Group’s blog. The opinions expressed in this blog post are the author’s only and do not […]
Medtech stories we missed this week: May 11, 2018
From ReShape submitting an approval application to Health Canada to Conavi receiving FDA clearance, here are seven medtech stories we missed this week but thought were still worth mentioning. 1. ReShape submits for Health Canada approval of ReShape balloon ReShape announced in a May 10 press release that it has submitted an application to the Medical Devices Bureau of […]
FDA clarifies multiple-function medical device oversight policy
By Stewart Eisenhart, Emergo Group New guidance from the US Food and Drug Administration addresses premarket authorization requirements for multiple-function products with at least one function that qualifies as a medical device. Get the full story here at the Emergo Group’s blog. The opinions expressed in this blog post are the author’s only and do […]