Shares in Eli Lilly (NYSE:LLY) fell today even though the drugmaker beat expectations on Wall Street with its second quarter results. The Indianapolis-based company posted profits of $1.01 billion, or 95¢ per share, on sales of $5.82 billion for the 3 months ended June 30, for bottom-line growth of 35% on sales growth of 8% compared […]
Featured
Brainstorm adds UCal Irvine Medical Center to late-stage NurOwn trial
Brainstorm Cell Therapeutics (NSDQ:BCLI) said today that it inked an agreement with the University of California Irvine Medical Center to enroll patients in the Phase III trial of its NurOwn mesenchymal stem cell-based platform for amyotrophic lateral sclerosis. This marks the third medical center to work with Brainstorm to enroll patients in the late-stage pivotal trial. […]
Study: Vaginal ring for HIV prevention safe in adolescent girls
A monthly vaginal ring that releases anti-HIV drugs was found to be safe and acceptable in an NIH-funded study of adolescent girls, according to data presented today at the 9th IAS Conference on HIV Science in Paris. The experimental ring boasted better adherence throughout the study compared to a daily oral tablet, the researchers reported. […]
India approves Anika’s Monovisc osteoarthritis treatment
Anika Therapeutics (NSDQ:ANIK) said today that regulatory authorities in India approved its single injection viscosupplement, Monovisc, as a therapy for pain associated with osteoarthritis in synovial joints. The Bedford, Mass.-based company’s product is already available in the U.S. Canada and Europe. Anika said it hopes to expand into Australia, New Zealand and other international markets in the […]
AntriaBio launches trial of once-weekly insulin
AntriaBio (OTC:ANTB) said today that the first patient was dosed in a Phase I first-in-human trial of its once-weekly basal insulin. The single ascending dose study is designed to test the safety, pharmacokinetics and dynamics of AB101 in patients with Type I diabetes. The second part of the study is slated to compare AB101 to Sanofi‘s […]
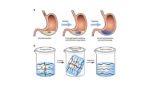
Responsive, ingestible drug-delivery material could boost patient compliance
Researchers from the Massachusetts Institute of Technology and Brigham & Women’s Hospital have developed a novel, responsive drug-delivery material that can reside in the stomach for up to nine days. The teams work was published today in Nature Communications. “One of the biggest issues in health care is noncompliance, people simply not taking their drugs,” co-senior […]
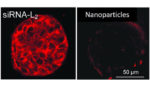
Study: Protein ferries molecule to tumors, triggering self-destruction
Ribonucleic acids, which can alter the behavior of cancerous cells, can be effectively ferried to tumors using a human protein called albumin, according to research conducted by a team of engineers at Vanderbilt University. The team’s work could help boost treatment for patients with triple-negative breast cancer, the researchers reported in the journal Proceedings of the […]
Sebacia raises $36m for microparticle acne treatment
Privately-held Sebacia Inc. said today that it closed a $20 million Series D financing round, led by existing investors Versant Ventures, Domain Associates, Accuitive Medical Ventures and Partners Healthcare Innovation Fund. New investors, including Salem Partners, also joined the round. The Duluth, Ga.-based company also said it closed a $16 million debt facility with Hercules […]
Takeda, BioSurfaces collab on GI drug-delivery devices
Takeda Pharmaceutical (TYO:4502) and BioSurfaces said today that the two companies inked a deal to develop novel medical devices intended to treat patients with gastrointestinal diseases. The deal brings together Takeda’s experience in the gastroenterology space and BioSurfaces’ method of developing nanomaterials from FDA-approved polymers for medical implants and drug-delivery devices. Financial terms of the […]
Abbott to study blood thinners after treatment with Xience drug-eluting stent
Abbott (NYSE:ABT) said today that it enrolled the first patient in a trial evaluating the short-term use of blood thinning drugs, called dual antiplatelet therapy, after implantation of the company’s Xience everolimus-eluting coronary stent. The 2,000-patient study is slated to assess if three months of DAPT is non-inferior to the current standard of 12 months […]