Horizon Therapeutics announced today that study results highlight the success of its Tepezza infusion for treating thyroid eye disease. Dublin, Ireland-based Horizon’s Tepezza proved in pooled Phase 2 and Phase 3 clinical trial data that it significantly improves proptosis (eye bulging) and diplopia (double vision) for TED patients in different subgroups, with most maintaining a […]
Clinical Trials
Genentech touts study results for spinal muscular atrophy drug in babies
Genentech today touted two-year data evaluating the use of its Evrysdi therapeutic in infants with symptomatic Type 1 spinal muscular atrophy (SMA). South San Francisco, Calif.-based Genentech’s Firefish Phase 2/3 global study for Evrysdi (risdiplam) in infants between 1-7 months old showed that the orally administered (liquid or feeding tube) therapeutic continued to improve motor […]
Innocoll Biotherapeutics initiates Phase 3 trials for collagen drug-device
Innocoll Biotherapeutics announced today that it initiated two Phase 3 trials to support a label expansion for its collagen drug-device implant. Athlone, Ireland-based Innocoll’s trials are seeking label expansion for the FDA-approved Xaracoll (bupivacaine hydrochloride) implant, according to a news release. INN-CB-024, a Phase 3, randomized, double-blind, placebo-controlled study is evaluating the efficacy and safety […]
Better Therapeutics initiates real-world study for digital therapeutic for diabetes
Better Therapeutics announced today that it commenced a real-world evidence study to evaluate its treatment for Type 2 diabetes. Collaborating with Steward Health Care, Better Therapeutics is evaluating BT-001, an investigational, prescription digital therapeutic (PDT) designed to deliver a novel form of cognitive-behavioral therapy to patients with uncontrolled Type 2 diabetes, according to a news […]
GH Research closes $125M oversubscribed Series B
GH Research Ireland announced that it closed an oversubscribed Series B financing round with proceeds of $125 million. Dublin, Ireland-based GH develops the GH001 therapeutic, a drug product for 5-MeO-DMT administration through a proprietary inhalation approach. A Phase 1 healthy volunteer trial showed GH001 to be well-tolerated while defining a dose range and individualized dosing […]
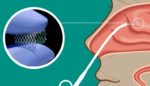
Lyra Therapeutics touts results from chronic rhinosinusitis treatment trial
Lyra Therapeutics (NSDQ:LYRA) presented positive results from a Phase 2 study of its therapeutic for treating chronic rhinosinusitis (CRS). Watertown, Mass.-based Lyra’s Phase 2 Lantern study evaluated its LYR-210 long-acting product for CRS, which is administered in-office to deliver a sustained release for up to six months as a non-invasive alternative to surgery for patients […]
TriSalus, University of Texas to collaborate on solid tumor treatment study
TriSalus Life Sciences and the University of Texas today announced a collaboration to evaluate the treatment of solid tumors. The Texas MD Anderson Cancer Center and TriSalus will collaborate on treatments for tumors of the pancreas and liver by integrating interventional delivery of SD-101, an investigational toll-like receptor 9 (TLR9) agonist, in combination with checkpoint […]
Nevro touts data supporting use of Senza system to treat diabetic neuropathy pain
Nevro (NYSE:NVRO) today touted results from a trial of its Senza system for treating painful diabetic neuropathy (PDN)-related chronic pain. Redwood City, Calif.-based Nevro, which published the results of the SENZA-PDN trial in JAMA Neurology, said in a news release that, if approved, the Senza spinal cord stimulation (SCS) system would be the first FDA-approved […]
BlueStar Genomics gains breakthrough nod for pancreatic cancer test
Bluestar Genomics announced today that it received FDA breakthrough device designation for its pancreatic cancer test. San Diego-based Bluestar’s proprietary noninvasive pancreatic cancer detection test is designed for use with patients with new-onset diabetes, according to a news release. The test uses a standard blood draw to assess whether an individual has an abnormal epigenomic […]
B. Braun, Infraredx to collaborate on drug-coated balloon catheter IDE study
B. Braun Interventional Systems (BIS) announced today that it is collaborating with Infraredx on a trial for the B. Braun SeQuent Please ReX. Bethlehem, Pa.-based BIS, a B. Braun affiliate, and Infraredx, a Nipro company, will work together to accelerate the FDA investigational device exemption (IDE) clinical trial for the SeQuent Please ReX drug-coated PTCA […]