Integra LifeSciences (NSDQ:IART) and Dr. Reddy’s Laboratories (NYSE:RDY) inked an exclusive distribution agreement for the DuraGen Plus and suturable DuraGen Dural Regeneration Matrices in India. According to the deal, Dr. Reddy’s will market and distribute the medical devices throughout India. The DuraGen Plus Dural Regeneration Matrix is indicated as a dural susbtitute for the repair of […]
Implants
Researchers win $1.8m NIH grant to develop implantable system for HIV prevention
Researchers at the University of North Carolina at Chapel Hill won a $1.8 million grant from the National Institutes of Health to develop an implantable drug delivery system for sustained HIV-prevention. The team said it plans to develop an injectable system that steadily releases pre-exposure prophylaxis over long periods of time. The injectable compound is […]
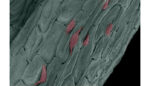
Study: Plants as 3D scaffolds for stem cells in biomedical implants
Researchers at the University of Wisconsin-Madison are aiming to create novel biomedical implants using 3D scaffolds made from the decellularized husks of parsley, vanilla and orchids. The team described their work in an article published in Advanced Healthcare Materials. “Nature provides us with a tremendous reservoir of structures in plants,” lead author Gianluca Fontana said in […]
Fibrosis: How to prevent it in medical device implants
Massachusetts Institute of Technology (MIT) and Boston Children’s Hospital researchers have discovered a way to prevent fibrosis from forming around medical device implants by blocking certain cells. The body’s immune system usually attacks implanted medical devices that are used for drug delivery, sensing or tissue regeneration. Defense cells in the body try to isolate the […]
Alimera, Brill Pharma ink exclusive distribution deal for Iluvien intravitreal implant
Alimera Sciences (NSDQ:ALIM) said today that it inked an exclusive distribution deal with Spain-based Brill Pharma for its Iluvien sustained-release intravitreal implant. According to the agreement, Brill Pharma will promote, market and commercialize the diabetic macular edema implant in Spain. It will also negotiate with the Spanish Ministry of Health on the implant’s public price and […]
Intersect ENT seeks FDA nod for Resolve steroid-releasing implant
Intersect ENT (NSDQ:XENT) said today that it submitted a New Drug Application to the FDA for its Resolve steroid-releasing implant to treat chronic sinusitis patients with recurrent sinus obstruction. The Menlo Park, Calif.-based company’s implant can be placed during a routine physician office visit and is designed to be a less invasive treatment option for recurrent […]
Study: Localized delivery of chemo improves prognosis of recurrent glioblastoma
Researchers from Amrita University in India demonstrated that localized and sustained delivery of a chemotherapeutic agent in brain tumors can improve the prognosis of recurrent glioblastoma. The team’s work was published in Scientific Reports. Although new drugs have been developed for glioblastoma multiforme, the overall survival rate for patients remains dismally low at 12 months, with […]
FDA asks Titan for more info before clinical trial of ropinirole implant
Titan Pharmaceuticals (NSDQ:TTNP) said today that the FDA put a hold on the clinical trial of its ropinirole implant and told the company to submit more information to the federal watchdog. After it completed an initial review of the implant’s Investigational New Drug application, the FDA asked Titan for final release test data on its ropinirole […]
Intersect ENT wins FDA nod for Propel Contour steroid-releasing implant
Intersect ENT (NSDQ:XENT) said today that it won FDA approval for its Propel Contour steroid-releasing implant for the treatment of chronic sinusitis in the frontal and maxillary sinsuses. The Menlo Park, Calif.-based company’s portfolio of steroid-releasing implants are used in patients undergoing ethmoid, frontal or maxillary surgeries to treat chronic sinusitis. “The approval of Propel […]
Alimera, Knight seek Canadian regulatory nod for Iluvien intravitreal implant
Alimera Sciences (NSDQ:ALIM) and Knight Therapeutics (TSE:GUD) said today that Health Canada accepted the companies’ New Drug Application for its intravitreal implant, Iluvien. The FDA approved Atlanta-based Alimera’s sustained release intravitreal implant in 2014 to treat diabetic macular edema in patients who have been previously treated with corticosteroids and did not have a clinically significant rise in […]