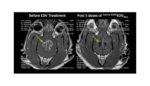
EnGeneIC said today that the FDA granted orphan drug designation to the company’s targeted EDV nanocells for the treatment of glioblastoma multiforme. The clinical stage biopharmaceutical company is developing bacterially-derived EDV nanocells as a nanoparticle, siRNA and miRNA delivery platform designed to target and kill tumor cells, while triggering the immune system’s natural anti-tumor response. […]
Nanoparticles
Study: Nanotechnology could cut dose of HIV treatment in half
Researchers from the University of Liverpool presented data this week evaluating the use of nanotechnology to improve drug therapies for HIV patients. The team touted its data at the Conference on Retroviruses and Opportunistic Infections in Seattle. The trial enrolled healthy patients and evaluated the use of orally-dosed solid drug nanoparticles to optimize HIV therapy, according […]
Azaya Therapeutics bought by Cytori, nanoparticle program launched
Cytori Therapeutics (NSDQ:CYTX) said yesterday that it completed its acquisition of San Antonio-based Azaya Therapeutics and its nanoparticle cancer drugs. In late January, San Diego-based Cytori said it planned to buy Azaya in a stock deal, offering Azaya shareholders $2 million in CTYX stock. Cytori also said that it paid $2 million upfront for costs associated […]
Virus shells target and deliver drugs to cancer cells
Researchers from Australia and the UK have developed virus-like nanoparticles to target and deliver drugs to cancer cells. The team’s work, which was published in the journal ACS Nano, showed that the particle could be produced in plants and that it can shuttle small molecules to cancer cells for targeted treatment. The team, lead by Frank […]
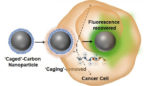
Researchers develop luminescent carbon nanodots to track drug delivery
Researchers from the University of Illinois at Urbana-Champaign have developed photo-luminescent carbon nanodots that exhibit reversible switching of their optical properties in cancer cells. The team’s work was published in the Journal of the American Chemical Society. The carbon nanoparticles could be applied to intracellular imaging and drug delivery, the authors said. “One of the major advantages […]
New technique rapidly tests nanoparticles for safety
Researchers from the University of Geneva and the University of Fribourg have developed a screening method that rapidly assesses and selects nanoparticles for their biocompatibility. The team’s work was published in the journal Nanoscale. The researchers touted that they can determine if a nanoparticle will be compatible with the human body in less than a week […]
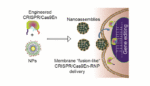
Researchers develop nanoparticle delivery system for gene-editing tool
Researchers from the University of Massachusetts Amherst have designed a nanoparticle delivery system to assist the gene-editing tool known as CRISPR/Cas9 across the cell membrane and into the cell nucleus, effectively avoiding the cell’s natural defense mechanisms. The team’s work was published in ACS Nano. “CRISPR has two components: a scissor-like protein called Cas9, and an […]
Aura Biosciences wins FDA nod for light-activated ocular melanoma treatment
Aura Biosciences said today that the FDA approved its Investigational New Drug Application for the company’s light-activated viral nanoparticle conjugates for the treatment of ocular melanoma. The federal watchdog’s approval allows Cambridge, Mass.-based Aura to move forward with initial clinical tests of AU-011. The Phase Ib open-label clinical trial, which is currently enrolling, is designed to […]
Acuitas touts lipid-nanoparticle delivery of mRNA vaccine for Zika
Acuitas Therapeutics touted preliminary data that evaluated its messenger RNA vaccine for the Zika virus. The company uses a lipid-nanoparticle delivery system to introduce its mRNA vaccine into cells. The team’s work, which was published in Nature, showed that a single low-dose immunization with its vaccine triggered an immune response in animals and protected them from infection at […]
Cerulean reviews clinical assets, nanoparticle-drug conjugates
Cerulean Pharma Inc. (NSDQ:CERU) said today that its board of directors launched a strategic review of its assests, considering alternatives for its clinical assets and nanoparticle-drug conjugate platform. Waltham, Mass.-based Cerulean said it could consider a sale, a merger, or an investment into the company. The board is also floating the possibility of a sale or […]